
The number of U.S. food manufacturing facilities inspected each year by the Food and Drug Administration has fallen by thousands over the last decade, despite Congress creating a mandate to increase inspections, an EWG analysis found.
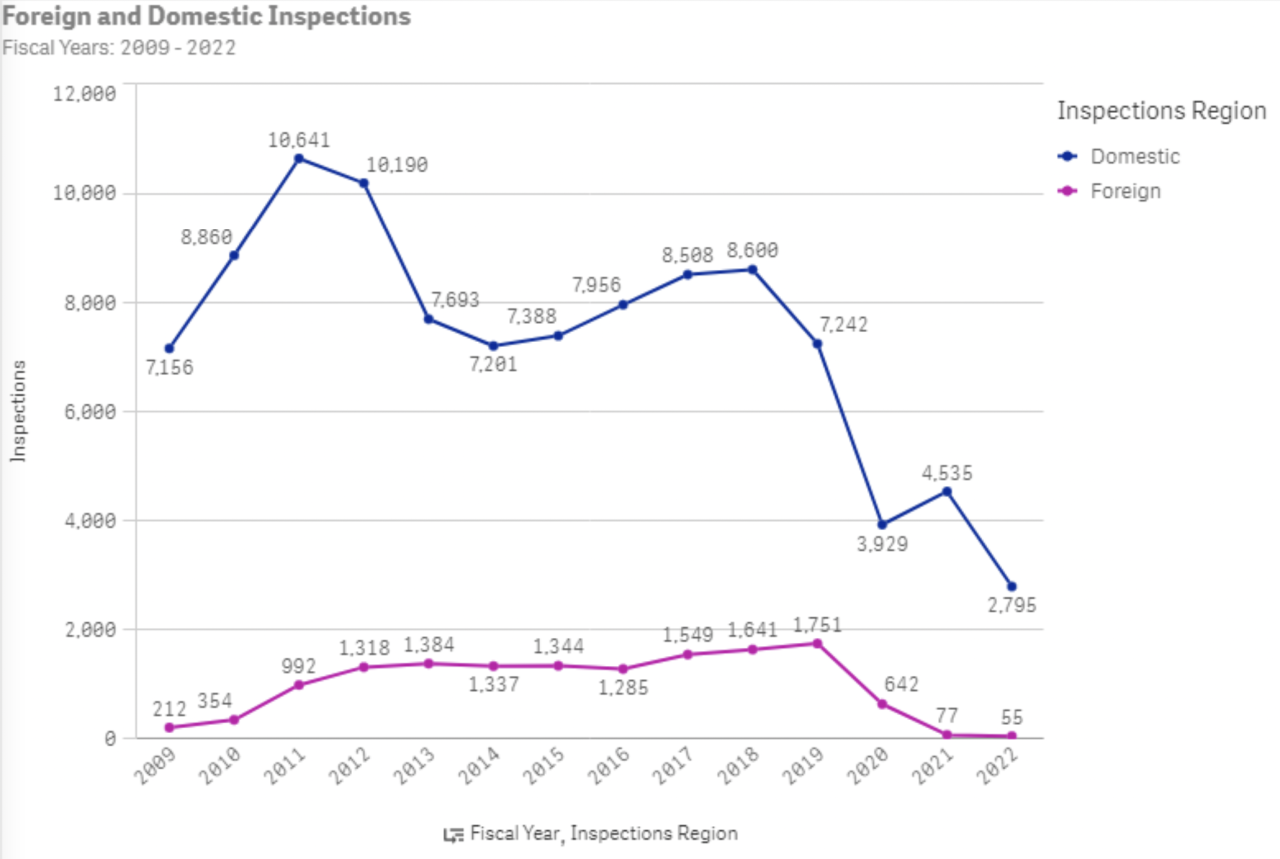
The FDA inspected just 4,535 U.S. food and cosmetics manufacturing facilities in 2021, compared to the 10,641 facilities it inspected in 2011, according to an EWG review of FDA data. So far, the FDA has inspected 2,795 facilities in 2022.
FDA combines inspections of food and cosmetics manufacturing facilities when reporting inspections, but most are facilities that make and process food.
Source: Food and Drug Administration
The number of inspections was falling even before the Covid-19 pandemic – despite a decade-old mandate from Congress. In 2011, Congress passed the Food Safety Modernization Act, or FSMA, which directed FDA to inspect high-risk facilities at least once every three years.
State inspections contracted by the FDA are also dropping, according to the Association of Food and Drug Officials, or AFDO. The total number of inspections by state officials on behalf of the FDA fell from 7,391 in 2019 to 6,131 in 2022, according to the AFDO.
The number of FDA inspections of foreign facilities increased slightly between 2011 and 2019, from 992 to 1,751, before plummeting during the Covid-19 pandemic, according to FDA data.
This week, FDA officials will testify to Congress on the agency’s failure to quickly inspect an infant formula facility in Michigan owned and operated by Abbott Laboratories, after learning one of the company’s powdered infant formulas could be contaminated.
The FDA waited months to inspect the Abbott facility after learning its formula could be contaminated. And after its inspectors confirmed the contamination, the FDA waited for weeks before alerting the public.
Since 2011, food outbreaks have gradually increased – at least until the pandemic dramatically changed our eating habits.
Why have inspections gone down?
As Congress finalized the FSMA, key legislators refused to support the user fees that typically fund FDA activities like drug and medical device reviews, even though food safety fees have the support of many food industry leaders. Since 2011, funding for other FDA centers that review drugs and devices has increased much faster than the one charged with keeping our food safe.
In addition, the Trump administration dismantled important reforms designed to make food safety a priority for the FDA. The Obama administration created a new position of deputy commissioner for food to oversee food safety. Rather than give the deputy commissioner even more power, especially over food safety inspections, the Trump FDA reversed the Obama-era decision. The Biden FDA has so far refused to make changes at the top of the agency, despite calls from states, industry, advocates and others to do so.


