HOME / daily use SPF / moisturizer with SPF

Mario Badescu The Moisture Magnet Sunscreen Lotion, SPF 15
Mario Badescu The Moisture Magnet Sunscreen Lotion, SPF 15

WHERE TO BUY
Health Concerns
-
MODERATECancer
-
HIGHAllergies & Immunotoxicity
-
MODERATEDevelopmental and Reproductive Toxicity
-
HIGHUse Restrictions
Efficacy Concerns
-
Caution We have flagged this product with 3 concerns Contains oxybenzone, a hormone disrupting chemical. Read more
Sunscreens can break down while still in the bottle. To be safe dispose of products when the mixture clumps or separates.
This product contains chemical active ingredient(s) that the FDA does not have enough health safety data to classify as safe and effective: AVOBENZONE , OCTINOXATE, OXYBENZONE. This product should not be used on kids younger than 3 years old.
-
moderateUVA/UVB Balance
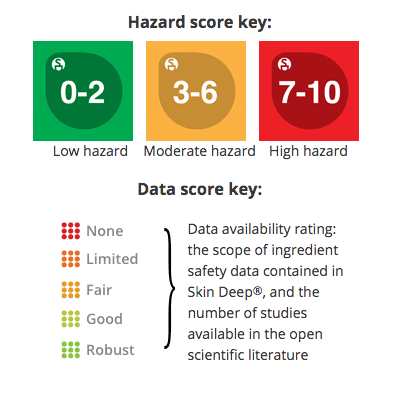
Ingredient Scores
Ingredients are scored based on their formulation and concentration in this product. Click on an ingredient for more information.

|
AVOBENZONE active ingredient 3%
Data Availability: Fair
|


|
||||
|
||||||

|
OCTINOXATE active ingredient 7.5%
Data Availability: Fair
|


|
||||
|
||||||

|
OXYBENZONE active ingredient 5%
Data Availability: Limited
|


|
||||
|
||||||

|
WATER
Data Availability: Robust
|


|
||||
|
||||||

|
BUTYLENE GLYCOL
Data Availability: Fair
|


|
||||
|
||||||

|
SESAMUM INDICUM (SESAME) OIL
Data Availability: Limited
|


|
||||
|
||||||

|
SACCHARIDE ISOMERATE
Data Availability: None
|


|
||||
|
||||||

|
MINERAL OIL
Data Availability: Fair
|


|
||||
|
||||||

|
ETHYLHEXYL METHOXYCRYLENE
Data Availability: Limited
|


|
||||
|
||||||

|
GLYCERYL MONOSTEARATE
Data Availability: Fair
|


|
||||
|
||||||

|
CETEARYL ALCOHOL
Data Availability: Limited
|


|
||||
|
||||||

|
TOCOPHERYL ACETATE
Data Availability: Limited
|


|
||||
|
||||||

|
STEARIC ACID
Data Availability: Fair
|


|
||||
|
||||||

|
DIMETHICONE
Data Availability: Fair
|


|
||||
|
||||||

|
GLYCERIN
Data Availability: Fair
|


|
||||
|
||||||

|
C10-30 CHOLESTEROL/ LANOSTEROL ESTERS
Data Availability: None
|


|
||||
|
||||||

|
GARDENIA FLORIDA EXTRACT
Data Availability: Limited
|


|
||||
|
||||||

|
ETHYLHEXYLGLYCERIN
Data Availability: Limited
|


|
||||
|
||||||

|
CETEARETH-20
Data Availability: Fair
|


|
||||
|
||||||

|
CARBOMER
Data Availability: Fair
|


|
||||
|
||||||

|
PHENOXYETHANOL
Data Availability: Good
|


|
||||
|
||||||

|
SODIUM HYDROXIDE
Data Availability: Good
|


|
||||
|
||||||

|
D&C Red No. 33 (CI 17200)
Data Availability: Fair
|


|
||||
|
||||||

|
Ext. D&C Violet No. 2 (CI 60730)
Data Availability: Limited
|


|
||||
|
||||||

|
CI 42090 (FD&C Blue No. 1 or D&C Blue No. 4)
Data Availability: Good
|


|
||||
|
||||||
Ingredients from label
Active Ingredients: Avobenzone 3%, Octinoxate 7.5%, Oxybenzone 5%; Inactive Ingredients: Aqua, Butylene Glycol, Sesamum Indicum (Sesame) Seed Oil, Saccharide Isomerate, Paraffinum Liquidum (Mineral Oil/Huile Minerale), Ethylhexyl Methoxycrylene, Glyceryl Isostearate, Cetearyl Alcohol, Tocopherol Acetate, Stearic Acid, Dimethicone, Glycerin, C10-30 Cholesterol/Lanosterol Esters, Gardenia Florida Fruit Extract, Ethylhexylglycerin, Ceteareth-20, Carbomer, Phenoxyethanol, Sodium Hydroxide, CI 17200 (Red 33), CI 60730 (Ext Violet 2), CI 42090 (Blue 1)