
Code names for untested chemicals, secret production amounts reported by unnamed companies, discharges of undisclosed amounts of pollutants – these occurrences are not the fantastical inventions of some Dr. Seuss book. These are realities currently allowed under the federal Toxic Substances Control Act, commonly referred to as TSCA, which became law in 1976.
The law has enabled the chemical industry to obscure its operations under a shroud of secrecy that ever since has hampered efforts to protect public health. Even though today’s advanced analytical chemistry techniques make it easier than ever for manufacturers to unlock the secrets of their rivals’ products through reverse engineering, the companies claim that they need to keep details about their products secret, and the law still allows them to do so.
This confidentially comes with a cost – it keeps the public in the dark about potential dangers and lets dangerous chemicals stay in the marketplace. By contrast, transparency often moves companies and regulators to take action to protect public health.
For example, EWG’s work on perfluorooctanoic acid, or PFOA – a toxic chemical once widely used in non-stick Teflon products – clearly demonstrates the importance of transparency and access to health and safety information. Internal DuPont safety testing documents on PFOA were only made public in 2002 as the result of a lawsuit against the company. The disclosures to the public and the Environmental Protection Agency led directly to the phase-out of the chemical.
In 2009, EWG called attention to the information black hole in TSCA that allows chemical names and production information to be claimed as trade secrets – formally “confidential business information” or CBI – and never made public. Within weeks of EWG’s report, the EPA took modest steps to control the most blatant overuse of the confidentiality stamp. Unfortunately, however, the EPA did not go far enough, and seven years later, little has changed. The lack of transparency remains a major roadblock to protecting public health from dangerous chemicals.
Updating its 2009 analysis, EWG has found that positive EPA actions have disallowed some CBI claims, but much more needs to be done. Today nearly two-thirds out of the 22,450 new chemicals manufactured since July 1979 are shrouded under cloak of secrecy. In the past six months alone, companies’ CBI claims have masked even the names of half the chemicals they reported to EPA as raising substantial safety concerns.
Congress is poised to legislate changes to TSCA this year for the first time in 40 years. Some of these changes will likely update the regulations that have permitted excessive and often unnecessary CBI claims. How meaningful these revisions will be, however, is largely dependent on the outcome of ongoing Congressional conference committee negotiations and the final language they adopt.
In the background, the chemical industry has been scrambling to protect its CBI claims. For the companies, preserving their power to use trade secret designations to cloak their products is a top priority, claiming that greater disclosure will hinder innovation. But the truth is that much greater transparency is possible without slowing research and development of new chemicals.
Introduction
Off the Books: EWG’s 2009 report
In 2009, EWG highlighted the remarkable extent to which the chemical industry can hide information about its products. Often omitted are the names of the chemicals and production data such as the identity of the manufacturer and the quantities produced, along with the names of chemicals in health and safety studies submitted to the EPA. EWG found that nearly two-thirds of all 17,000 chemicals added to the TSCA inventory since 1979 had been claimed confidential, including substances linked to cancer and hormone disruption. The EWG report also found that hundreds of chemicals produced in significant volume were claimed confidential, including 10 intended for use in children's products.1
EPA actions on Confidential Business Information since EWG’s report
Three weeks after EWG’s Off the Books report was published, EPA announced it would make major policy changes to ensure that CBI be claimed only in appropriate circumstances. The agency said that it “planned to reject CBI claims for chemicals submitted to EPA with studies that show a substantial risk to people's health and the environment and that have been previously disclosed on the TSCA Chemical Inventory.”2
In 2010, EPA announced a number of changes to its handling of CBI claims, including:
- A voluntary challenge to the major industry trade associations asking them to declassify outdated CBI filings and to greatly limit future CBI claims.3
- A proposed rule that would begin to review CBI claims on “chemical identity” in health and safety studies submitted to EPA. In its notice, the agency cited TSCA’s Section 14(b) – which prevents companies from claiming confidentiality in cases when a chemical formula is not directly disclosed publicly.
- A policy announcement that chemical identity would be considered an integral part of health and safety studies and could not be hidden under CBI claims. Specifically, EPA announced that:
Section 14(b) of TSCA does not extend confidential treatment to health and safety studies, or data from health and safety studies, which, if made public, would not disclose processes used in the manufacturing or processing of a chemical substance or mixture or, in the case of a mixture, the release of data disclosing the portion of the mixture comprised by any of the chemical substances in the mixture. Where a chemical identity does not explicitly contain process information or reveal portions of a mixture, EPA expects to find that the information would clearly not be entitled to confidential treatment.4
This last provision met with significant opposition from the chemical industry. The American Chemistry Council submitted a white paper to the Administration arguing that EPA’s interpretation of the TSCA statute was incorrect: “Congress intended for EPA to protect chemical identities in submitted health and safety studies while also providing the public with the health and environmental information it needs to evaluate those studies. In other words, EPA must balance the competing interests, as it has done for nearly 30 years.”5 The official stance of the Board of Governors of the Society of Chemical Manufacturers was: “chemical identity is not an essential element of health and safety studies and must continue to be protected against public access.”6 To date EPA’s general announcement has not resulted in a general policy of publishing the name of chemicals claimed confidential in health and safety studies.
Testifying in 2013 before the House Subcommittee on Environment and the Economy of Committee on Energy and Commerce, the Government Accountability Office (GAO) reasserted its recommendation that EPA routinely challenge CBI claims in order to “expand public access to this (chemical) information – such as sharing it with state environmental agencies and foreign governments, which potentially limits the effectiveness of these organizations’ environmental risk programs.”7
In a 2014 update to its CBI declassification initiative, the EPA reported that since 2009 it had:
1) declassified 643 previously confidential chemical identities and moved them to the public TSCA Inventory;
2) increased the number of publicly available health and safety studies on chemicals listed in the TSCA Inventory;
3) completed reviews of more than 24,000 declassification cases; and
4) reduced the number of TSCA CBI cases that needed to be reviewed for relevancy from 22,483 to 565.8
EPA uses its ability to review certain CBI chemicals and if applicable, disallow these claims if they don’t meet EPA’s “substantive criteria in the (TSCA) statute.”9
In light of these EPA actions and the imminent prospect of legislative changes, EWG has taken a fresh look at the issue of confidentiality and TSCA.
Has anything changed?
The Good
- Confidentiality claims masking chemical identity in 7,690 industry production volume reports dropped from 1,105 in 2006 to 230 in 2012.
- In 2006, companies used CBI claims to shield the identities of at least 58 of 215 chemicals intended for use in children’s products. As of 2012, only one of 355 children’s use chemicals submitted to EPA claimed CBI.
- The chemical inventory is now freely available on the EPA website.
- EPA published guidance documents for substantiating CBI claims submitted in companies’ production information reports.
The Bad
- The public has no access to the name (technically, the specific chemical identity) of approximately 17,500 of the more than 85,000 chemicals on the master TSCA inventory compiled by EPA.
- Industry has placed CBI claims on the identity of 13,975 new chemicals introduced since 1979 – nearly two-thirds of the 22,450 chemicals added to the TSCA list in the past 40 years.
- In the last six months (since October 2015), chemical identities were claimed secret in over 50 percent of industry’s more than 250 Section 8(e) submissions. Under TSCA’s section 8(e), companies are required to hand over all documentation showing that a chemical presents “a substantial risk of injury to health or the environment.” By definition, Section 8(e) filings raise the greatest health concerns.
- In 2014, EPA published an update to its 2012 Chemical Data Reporting (CDR)– which showcases chemicals submitted to the TSCA Inventory that are produced in amounts greater than 25,000 pounds a year in a single facility. Of the 33,000 submissions to the CDR, more than 14,000 (44 percent) had at least one CBI claim.
- 3,379 (10 percent) had CBI claims on the company’s name.
- 3,591 (11 percent) had CBI claims on the production site.
- 9,915 (30 percent) had CBI claims on the amount of a particular chemical manufactured at a particular site.
TSCA
TSCA is the primary federal law regulating toxic chemicals in the United States. It is the only major federal environmental statute that has not undergone significant updates or amendments in the 40 years since.
The law is lengthy and complicated, but broadly its focus is chemical management and safety. TSCA created an inventory of chemicals in commerce and a process for notifying EPA when new chemicals were introduced.
TSCA grandfathered the 60,000 chemicals already in commerce in 197910, exempting them from the notification process. For those chemicals, the law gave the EPA authority to collect safety information or require new testing. It also authorized EPA to analyze the existing chemicals for safety and to adopt regulations or ban them as necessary. Other key provisions address trade secret protection, state chemical regulations, penalties and fees.
There is a consensus that TSCA has failed to ensure chemical safety. Although EPA receives notifications for newly introduced chemicals, they often come with little to no safety data and are allowed to go on the market without any safety review or explicit EPA approval. Bureaucratic obstacles make it difficult for EPA to get data on existing chemicals on the market and even harder to regulate them. Of the 60,000 chemicals grandfathered in 1979, EPA has only regulated five chemicals using their authority to protect against unreasonable risk. The law is so ineffective that EPA was stymied when it attempted in 1989 to ban deadly asbestos.
One of the law's greatest weaknesses, as documented by EWG’s 2009 “Off the Books” report, is that chemical companies have wide leeway to keep information about their products secret. With few exceptions, companies can claim that most information about their chemicals is a “trade secret.” There are no minimum requirements for making a trade secret claim, the protection never expires and the information does not have to be shared with state and local authorities. Keeping the information secret makes it harder for the EPA, state and local governments, academic scientists and public health researchers to assess whether chemicals in commerce are safe.
The TSCA Inventory
When TSCA was enacted in 1976, EPA proceeded to gather information about chemicals known to be in use at the time. This list, known as the TSCA inventory, includes basic information such as: the preferred chemical name; any synonyms; its molecular formula; whether it was processed by EPA’s new chemical program; and whether the chemical is regulated under TSCA. Since enactment, EPA has added chemicals to the inventory every time a new one goes into commerce. While this creates an impressive list, little has been done to track which chemicals are actually used.
EPA has failed to fulfill its statutory requirement to maintain and “keep current”11 its list of chemicals in commerce. Unused or obsolete chemicals are not removed from the inventory. Over the last four decades the inventory has evolved to be just like Hotel California in the popular song by that name: “You visit any time you want but you can never leave.”
As a result, the inventory has grown into an unwieldy list of questionable value.
Many of the chemicals originally on the inventory have likely been phased out and replaced. In part because of EPA’s inaction and in part because of trade secret protections, it is nearly impossible to know which of the 85,000 chemicals now on the inventory are still being used and may be ingredients in consumer products. In 1979, after years of collecting data from manufacturers, the EPA estimated that there were 60,000 chemicals active in commerce, with 1,800 (3 percent) claimed as confidential trade secrets.12
EPA announced a proposed rulemaking in 2008 to reset the inventory to include only chemicals that were in use at the time. This effort fizzled, however, and in 2010 EPA’s Director of the Office of Pollution Prevention and Toxics (OPPT), Wendy Cleland-Hamnett, told a meeting organized by the chemical industry that the agency was no longer seeking an inventory reset.13
The Senate version of the pending legislation to revise TSCA would address the problem of EPA’s inaction, but the House version is silent on the issue.
New Chemicals
Following the publication in 1979 of the initial TSCA inventory list, the law required that manufacturers notify EPA of all new chemicals before they were added to the inventory of chemicals allowed for use:
“Pre-manufacture notices” (PMNs) must be submitted to EPA 90 days prior to producing or importing a chemical not already in the TSCA Inventory.”14
These notices are not required to include any health or safety information, and often none is provided. EPA only takes action to restrict or regulate about 10 percent of chemicals in the notices,15 which could be attributed to the absence of any safety information.
The public inventory lists only basic information – such as the chemicals registry number, preferred name and molecular formula. A chemical’s synonyms, former registry numbers and EPA rule-making flags on a given chemical can be listed as well, but are not required. The confidential inventory includes chemicals that are submitted with confidential business information, or CBI claims and lists only those chemicals’ generic names, registry number and regulatory flags. What is not required in either lists are a chemical’s manufacturer, manufacturing location(s), production values, type of products the chemical is typically used in and any updates to manufacture or use including whether it is still being manufactured.
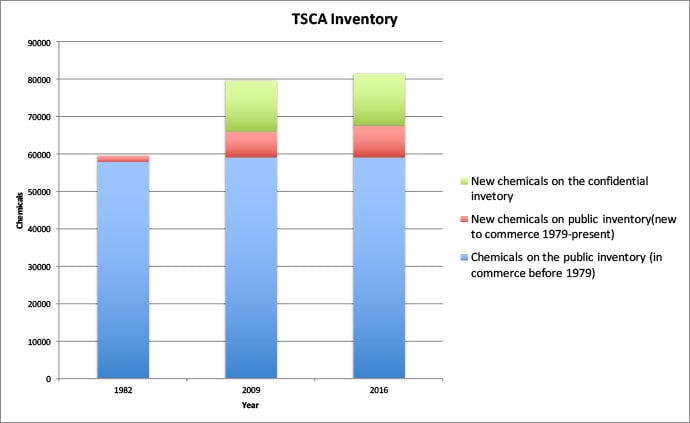
EWG’s analysis of the TSCA inventory as of March 2016 inventory found that the total number of chemicals listed increased from the original 60,000 to more 85,000 chemicals. The public inventory provides unique identifying information on just 67,634 chemicals. Of these, 8,475 are identified as having been added after publication of the inventory in 1982. There are an additional 13,975 confidential chemicals submitted under the new chemicals process that are listed only under a generic name and a generic “accession” number.
EWG’S 2009 analysis showed that the identities of two-thirds of the chemicals introduced since 1979 were confidential. There has been no significant change since then.
Confidential Claims on Harmful Chemicals
It is especially important to limit confidentiality claims on chemicals that pose substantial risks. However, that is not the case under current law. Section 8(e) of TSCA gives EPA the ability to collect hazard studies on both existing and new chemicals. It also requires companies to notify EPA when they obtain information that shows that a chemical or mixture “presents a substantial risk of injury to health or the environment.”16
These Section 8(e) submissions are critical for flagging harmful chemicals already in commerce as well as emerging concerns, but companies can tack confidential trade secret claims onto these submissions, undermining their usefulness for researchers and others.
Of the more than 250 Section 8(e) submissions in the six months since October 2016, more than half carried CBI claims masking the chemical’s specific name.17 Section 8(e) chemicals are those that raise the greatest concern, and shielding them with confidentiality claims can slow or block actions to protect human health.
For example, one Section 8(e) submission from February 2016 – reported that an unnamed chemical from an unnamed company caused decreased fertility and changes in the lungs, spleen, stomach, intestines and vagina. The test substance also killed half the rats exposed to a 100/80 mg/kg/day (milligrams per kilogram) concentration.18
In another 8(e) submission from November 2015 – several pregnant test subjects received 5mL/kg dosages of a chemical daily. All pregnant test subjects survived the testing, but had significantly reduced weight fetuses, which was concluded, “to be a test-item related effect.”19 The National Institute of Health states low birth weight babies “may become sick in the first days of life or develop infections. Others may suffer from longer-term problems such as delayed motor and social development or learning disabilities.”20
If those chemicals killed subjects or caused reproductive harms in lab tests, you would think researchers and public health advocates would have the right to know about it. But because the manufacturer claimed the chemical identity was a trade secret, the EPA can only release the redacted studies and generic identification — leaving researchers, public health advocates and others in the dark as to which specific chemicals produced these alarming effects.
Chemicals Produced in Significant Volume
As a complement to the TSCA Inventory for the most commonly used chemicals, EPA implemented the Inventory Update Reporting (IUR) program in 1982. It requires that companies that produce or import a chemical in quantities over 25,000 pounds a year at a single facility submit general information to EPA once every five years – including where it was produced, how much was produced and who produced it.21 Since the first reports in 1986, there have been five reporting periods and snapshots of data collected by EPA. There have been a few changes to reporting requirements but the number of chemicals reported under the system have remained close to 8,000 in each reporting period.22
In 2011, EPA renamed the program Chemical Data Reporting (CDR). Under it, companies producing or importing at least 25,000 pounds a year of a chemical at one facility must collect information annually and report once every four years. The latest CDR update lowered the reporting threshold for use and processing information from 300,000 pounds a year to 100,000.23 In the first reports submitted under this threshold in 2012, 1,626 companies listed 7,690 chemicals produced or imported at 4,785 sites.24
The 2012 CDR report detailed under what circumstances a business can place a confidentiality (CBI) claim on a chemical and how the EPA would substantiate those claims.
“Submitters may designate individual CDR data elements as CBI when they report information. However, chemical identity may only be claimed confidential if the chemical is listed on the confidential portion of the TSCA Inventory. Processing and use data elements can be claimed as CBI if a manufacturer (including importer) believes that the release of information will reveal trade secrets or confidential commercial or financial information. Submitters are required to provide upfront substantiations of confidentiality claims for chemical identity, site identification, and processing and use information by answering a series of questions in the reporting form. A blank response or a response that is designated as ‘not known or reasonably ascertainable’ may not be claimed as confidential.”25
Under the IUR program, the EPA initially provided very little information on how it regulated confidentiality claims. The 2012 CDR report said 27 percent of submissions had claimed confidentiality on the chemicals’ use or processing information.
For its 2009 “Off the Books” report, EWG requested information on how many IUR (now CDR) chemicals had been listed as confidential throughout the program’s history. EPA responded that in 1990, there were 261 confidential IUR chemicals. By 2006, that number had skyrocketed, with 1,105 IUR chemicals shielded by confidentiality claims.26
Just before publication of EWG’s 2009 analysis, the agency revoked the confidential status of 530 of the 1,105 IUR chemicals, lowering the total number of confidential, widely produced chemicals down to 575.
EPA continued reviewing CBI claims on chemicals produced in volumes over 100,000 pounds a year, and its 2012 report on the CDR program revealed the the number of confidential claims on medium- and high-production volume chemicals was down to 230. Because of changes in what EPA regulated and reported under the two programs, it is hard to directly compare the IUR and CDR data.
Of the more than 33,000 submissions to the 2012 CDR database, over 14,000 (44 percent) carried at least one confidentiality claim. This number is larger than the CDR list of 7,690 chemicals because it takes into account the numerous sites and companies that produce the same chemicals.27
Pending TSCA Bills
Congress passed two bills last year to update TSCA: H.R. 2576, the TSCA Modernization Act, and S. 697, The Frank R. Lautenberg Chemical Safety for the 21st Century Act. Both seek to address the issue of chemical trade secret claims to varying degrees. S. 697, the Senate bill, also takes steps to update the TSCA inventory to give an idea for the first time which of the 85,000 chemicals on the inventory are actually in use.
Confidential business information
Both the House and Senate bills address the issue of trade secrets but take very different approaches.
Health and safety studies.
Under current law, one major exception to trade secret protection is that EPA is allowed to release information in health and safety studies about a chemical. Both bills retain a form of this exemption, with some key differences.
The House bill rolls back current law by allowing companies to protect “data that discloses formula (including molecular structure)” in their health and safety studies, which would likely allow them to keep the chemical identity secret. Under current law, the chemical identity cannot be claimed as a trade secret in health and safety studies. The Senate bill does not include this rollback.
Additionally, the Senate bill strengthens the framing of this exception by saying that information from health and safety studies “shall not be protected” from disclosure. The House bill retains the softer approach in current law by stating that the EPA “shall not prohibit” disclosure of information – leaving the ultimate decision up to EPA discretion.
Information for state and local regulators.
Under current law, EPA is not able to share trade secrets with state and local regulators, even to enforce local laws. The House bill would give EPA discretion to share this information with state and local regulators. The Senate bill would make it mandatory to share the information.
Requirements for claiming trade secret protection.
The Senate bill provides clear requirements for what companies must do to claim trade secret protection, including showing that the information could not be obtained by reverse engineering. Like current law, the House bill does not provide any such guidance.
Trade secrets in case of a ban or phase-out.
Under the Senate bill, a chemical loses trade secret protection if the EPA bans or phases it out – an improvement over current law. There is no corresponding provision in the House bill.
Review and re-substantiation.
Under current law, trade secret protection status never expires. Under both bills, new trade secret claims would expire after 10 years, after which the claims would have to be re-substantiated in order to be renewed. However, only the Senate bill would require EPA to look systematically at past trade secret claims and require that they be dropped or renewed. Under the House bill, past claims would remain secret indefinitely.
The House bill would also only allow EPA to seek re-substantiation for trade secret claims when those claims are about to expire. By contrast, the Senate allows or requires re-substantiation whenever safety concerns arise. EPA would have discretion to request re-substantiation if a chemical is designated as high priority for safety review or the agency determines that more information is needed to make a safety determination. Re-substantiation would be required anytime a chemical is found to pose serious risks or if a chemical that had been out of use is put back into commerce. The Senate bill’s triggers for review and re-substantiation would provide an important check on chemical secrecy.
One other important difference between the House and Senate bills involves the treatment of the TSCA inventory. The House bill would not change the TSCA inventory, but the Senate bill would require industry to notify the EPA when a chemical is no longer used or becomes inactive. Dividing the TSCA inventory into active and inactive lists would greatly help the EPA and other researchers to identify exactly which chemicals they should focus on.
Recommendations
Instead of ensuring accountability from chemical producers, the existing system protects chemicals from disclosure and possible public review though excessive use of confidentiality claims.
The chemical industry has a tight grip on the information needed to implement the most basic protections from chemicals that can affect our health and environment.
Since EWG first reported on the issue of trade secrets in 2009, EPA has taken the initiative to limit the flood of CBI claims on new chemicals added to the TSCA Inventory as well as revoking decades-old CBI claims. However, more must be done.
Important steps to improving transparency under TSCA include:
- As a general rule, the identity of chemicals in health and safety studies should be made public when it enters commerce.
- The TSCA inventory should be regularly updated to reflect the chemicals actually in commerce.
- All CBI claims should be re-substantiated regularly.
- The more health protective language in the Senate bill should be used in the final legislation.

