
The unique bond between a mother and daughter starts in the womb and evolves over a lifetime, as each adapts and grows with the other in an elaborate interplay of nature and nurture. Shared bonds of common genetics and a common environment — their home, the air they breathe, and the food they eat — inextricably link daughters and mothers. Now, new laboratory tests of mothers and their daughters show that these same two facets of nature and nurture — genetics and environment — combine to create another, unwanted aspect of the ties that bind: a common body burden of industrial chemicals.
Tests commissioned by the Environmental Working Group of four mothers and their daughters found that each of the eight women's blood or urine was contaminated with an average of 35 consumer product ingredients, including flame retardants, plasticizers, and stain-proof coatings. These mixtures of compounds found in furniture, cosmetics, fabrics, and other consumer goods, have never been tested for safety.
These and many other chemicals are building up in the bodies of all Americans. But EWG's tests found three eye-opening trends about pollutants that can pass through a mother's placenta or breast milk into her daughter's body:
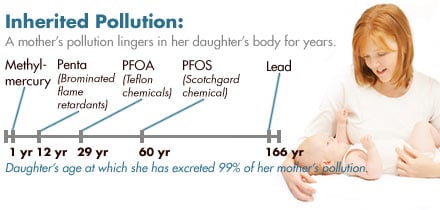
- A substantial portion of the chemical burden inherited at birth by the daughters in this study will last for decades; some will last a lifetime. In turn, the daughters can pass on to their children some of the same molecules of industrial chemicals that they inherited from their mothers. The mothers in this study, however, probably received far less inherited pollution at birth. Instead, they accumulated their pollution over a lifetime, as members of the first generation of parents routinely exposed to commercial chemicals like flame retardants, plasticizers, and stain repellants. The estimated age by which a daughter will purge 99 percent of the inherited pollution found in this study ranges from one day for phthalate plasticizers, to one year for mercury, to between adolescence and 60 years for common flame retardants and stain-proofing chemicals, to longer than a lifetime, 166 years, for lead. And while each daughter begins life with an inherited load of pollution, she also continues to build up additional pollutants through childhood and beyond.
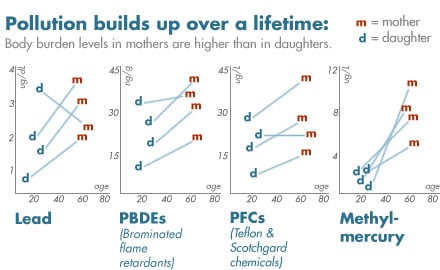
- Chemicals that persist in the body were found at higher levels in mothers than daughters, showing that chemicals can build up in the body over a lifetime, posing long-term, persistent health risks. Mothers harbored in their bodies an average of 1.5 to 5.2 times more pollution than their daughters for four chemicals or chemical families: lead, methylmercury, brominated flame retardants, and the Teflon- and Scotchgard-related perfluorochemicals known as PFOA and PFOS.
The mothers and daughters in this study join 64 other people tested in six EWG biomonitoring programs conducted between 2000 and 2006, including a study published in July 2005 of 10 newborn babies with an average of 200 industrial pollutants, pesticides, and other chemicals in each child (EWG 2005). In total, EWG biomonitoring has found 455 different pollutants, pesticides, and industrial chemicals in the bodies or cord blood of 72 different people. By any measure this is an astounding number of contaminants — a burden of pollution that is made even more troubling by the lack of health studies or safeguards for their individual or combined toxic effects.
The pressing need for change. Exposures in early life heighten concerns over health risks from these exposures. EPA studies show that children from birth to age two are 10 times more sensitive to carcinogens than are adults (EPA 2005). A new study from UC Berkeley shows that children may be up to 164 times more sensitive than adults to neurotoxic pesticides called organophosphates (Furlong et al. 2006), and innovative research from Washington State University demonstrates that chemicals can confer toxicity four generations after exposure, by forcing permanent, heritable changes in gene expression that can change the body's ability to metabolize and excrete toxic chemicals (Anway et al. 2005).
But even in the face of growing evidence of health risks from chemicals, particularly for children, the government has been slow to act, as demonstrated by three important, pending federal and state initiatives:
- National Children's Health Study. In a recent act of irony, the Bush administration presented the well-deserved, prestigious "Children's Environmental Health Champion Award" to Dr. Philip J. Landrigan, a longtime children's health researcher and advocate from New York's Mt. Sinai School of Medicine, just months after submitting a budget to Congress that would cut off funding for the seminal children's health study he was to spearhead. This pivotal project aims to track health and chemical exposures for 100,000 children from birth to age 21, and would dramatically expand our understanding of children's environmental health. Research like the now jeopardized Children's Health Study would yield the powerful, raw data needed to spur significant advances in scientific understanding of the health risks from childrens' inherited and accumulated exposures.
- California Biomonitoring Program. The California Legislature is considering a bill, SB 1379, that would establish the first state program to track pollutants in people. The program would help state health officials better understand the relationship between chemical exposures and increasing rates of disease, track the impact on public health of emerging chemicals of concern, and identify communities disproportionately impacted by chemical exposures. Gov. Arnold Schwarzenegger vetoed a similar bill last year, and the chemical industry is again lobbying furiously to kill it. But proponents believe the emerging science on chemicals in the human body, and Californians' overwhelming support for the right to know what chemicals are in their bodies, are bettering their chances this year.
- Federal Kid Safe Chemicals Act. In 2005 Congress responded to the science of body burden with a bill that would fill many of the gaps that leave public health at risk when it comes to chemical exposures. The Kid Safe Chemicals Act (S1391 and HR 4308), the first major update of 1976 Toxic Substances Control Act, was introduced in the Senate and House in July and December 2005, respectively. This dramatic rewrite of industrial chemical law would require companies to document the safety of chemicals before they go on the market, and, most importantly, would require that chemicals be safe for the fetus, infant, and other vulnerable populations — a simple, commonsense idea completely absent from current federal law.
All of these measures, if passed and implemented, would dramatically advance our understanding of health impacts from chemical exposures, and would go a long way toward sealing the gaps that leave mothers, daughters, and others at risk from the human body burden of industrial chemicals.
Findings
Chemicals in mothers and daughters. In November and December 2005, four mothers and their daughters donated blood and urine to a biomonitoring program, collecting a first morning void of urine, and rolling up their sleeves for a needle that collected blood into 10 small glass and plastic tubes with color-coded tops. From tests of these fluids they would not learn of their cholesterol, liver function, or thyroid hormones. Instead, they would uncover personal facts more unique and far more difficult to diagnose: the levels of 70 industrial chemicals circulating in their bodies at the moment the fluids were released, mixtures that have never before been tested in combination, carrying health risks that are largely unknown.
Together, the body burdens of four mothers and their daughters include seven plasticizers called phthalates (pronounced tha'-lates) used in plastic, cosmetics, and building materials; chemicals known as bisphenol A (BPA) and BADGE-40H, which are, respectively, residues from hard plastic bottles and from epoxy resins used to line food cans; a suite of brominated flame retardants used in computers, TVs, and foam furniture; perfluorochemicals related to Teflon and Scotchgard; and lead and mercury, toxic metals from chipping paint and tainted seafood.
The data are remarkably consistent with two tenets of the human body burden of industrial chemicals: levels of persistent chemicals, those that cling to body tissues for decades, build up to higher and higher levels in the body over a lifetime; while levels of non-persistent chemicals, those that pass through the body over a few days' time, vary widely and depend not on as much on age as on day-to-day contact with everyday consumer products.
Below we describe findings from this investigation, that shows for four mothers and their daughters what it means to share and inherit the body burden of industrial chemicals.
The pollution we inherit. Among the eight mothers and daughters tested, we found that an average of 93 percent of the chemicals daughters harbor in their body tissues and fluids are also found in their mothers, compared to just 81 percent held in common between daughters and 16 other women who were tested. This difference was statistically significant in three of four daughters, and emerged despite the fact that many chemicals in the study are expected to be found in most people tested, regardless of family connections. Included in this study are chemicals that rate among the most ubiquitous pollutants ever detected in people, including phthalate plasticizers, lead, mercury, brominated flame retardants, and the Teflon and Scotchgard chemicals known as PFOA and PFOS.
Studies show that a mother confers a portion of her chemical body burden to a child in the womb (EWG 2005), and continues to pass down pollution after birth through her breast milk. Studies have found that a mother passes to her child an estimated 50 percent of her own body burden of persistent chemicals through six months of breast feeding (LaKind et al 2000). The undeniable phenomenon of inherited pollution has led researchers to note that nursing a child is "the only known way" in which a women can unload from her body "large amounts" of persistent chemicals (Jorgenson 2001). Nevertheless, child health advocates are quick to note that breast milk remains the best food for babies, despite the pollution: "The advantages of breast-feeding have been documented in the neonatal period and extend throughout childhood and into adulthood" (Solomon and Weiss 2002). That no substitute for a mother's milk has been found raises deep concerns about the ubiquity of pollution in this otherwise singularly perfect infant food.
Inherited pollution that lasts a lifetime. A daughter's birth load of inherited pollution can last a lifetime, and a daughter can act as a conduit from grandmother to granddaughter, passing inherited chemicals to her own children during pregnancy and nursing. While the daughters in this study excreted an average of 99 percent of their inherited load of methylmercury by their first birthdays, similar levels of purging don't occur until adolescence, childbearing years, senior years and beyond for other pollutants even more persistent in the body, including flame retardants, Teflon chemicals, and lead. And of course, for these daughters, new exposures continue after birth and after giving birth; many of the chemicals included in this study continue to accumulate over a lifetime.
At one end of the spectrum are the plasticizers called phthalates, which pass through the body over the course of a day. Mercury is more persistent: children excrete 99 percent of their inherited load of methylmercury, on average, by their first birthday, though within that relatively short span the chemical may already have exerted its damaging effects to the developing brain. Through their natural excretion processes, daughters in this study would not reach this level of purging for brominated flame retardants and Teflon and Scotchgard chemicals until they reach ages estimated to range from adolescence to 58 years of age. And unless they live to be 166, these daughters could not rid themselves of 99 percent of the lead passed through the placenta into their bodies in the womb.
The daughters in this study range in age from 17 through 29. The science of chemical persistence tells us that at their current ages they continue to hold in their bodies, on average, an estimated 60 percent of the lead, four percent of the Teflon chemical PFOA, and 19 percent of the Scotchgard chemical PFOS passed to them from their mothers.
At age 29 at the time of sampling, the oldest daughter in this study would harbor an estimated one percent of the original mass of the Teflon chemical known as PFOA and half of the lead passed to her from her mother across the placenta. But last fall she passed a portion of this pollution on to her newborn daughter; her own loads of pollution are likely lower now. The youngest daughters in this study, at age 17, continue to store an estimated 68 percent of the lead passed to them in utero from their mothers, and 27 percent of the Scotchgard chemical PFOS.
These estimates are derived from studies which indicate that the body can rid itself of half of any given dose of mercury, PBDE-47, Teflon (PFOA), Scotchgard (PFOS), and lead in two months, 1.8 years, 4.3 years, nine years, and up to 25 years, respectively (NAS 2000, Geyer et al. 2004, Burris et al. 2002, Olsen et al. 2003, Lin et al. 2004).
An unwanted inheritance that accumulates over a lifetime. Levels of persistent brain toxins and carcinogens are higher in mothers than in their daughters. In 14 of 16 cases, mothers' bodies held higher amounts of four body-persistent chemicals or chemical families than their daughters' bodies, including lead, methylmercury, stain- and grease-resistant chemicals in the Teflon family, and brominated fire retardants known as PBDEs. The two exceptions to this trend were found in the relatively low levels of lead and perfluorochemicals found in a mother who had undergone detoxification procedures to rid her body of chemicals three weeks prior to giving blood and urine for this study.
The majority of these compounds persist in the body for years, and build up over time, a chemical property that accounts for higher levels in mothers relative to their daughters. But a mothers' higher levels can stem from a wide variety of other factors as well, including differing exposures (higher seafood consumption in mothers, for instance, can increase methylmercury pollution); higher income, which can drive higher rates of purchasing new products with flame retardants and perfluorochemicals; and physiologic differences like a diminished ability to excrete chemicals (Geller and Zenick 2005).
Just as in childhood, the later years in life are a time of susceptibility to chemical toxicity. As the body ages, it becomes not only more vulnerable to industrial chemicals, but also less efficient at metabolizing and excreting these chemicals.
As they age people can be exposed to higher amounts of some chemicals. Exposure patterns change — for instance, older adults spend more time indoors than younger adults, and during this time can be exposed to the many hazardous air pollutants found at higher concentrations indoors than outdoors. Changes in physiology with age can increase the amount of a chemical that is absorbed into the body. For example, as the skin ages, it allows more of some chemicals to penetrate into the blood vessels below.
With age also comes a reduced capacity to metabolize and excrete chemicals. EPA scientists summarize a number of these phenomena in a recent review of aging and susceptibility: decreased muscle mass and increased fat stores result in higher accumulation and reduced excretion of some chemicals; kidney function decreases with age; liver activity drops, with an accompanying potential for slowed detoxification of chemicals and reduced excretion rates.
Aging leads to physical changes that increase susceptibility to chemicals. For instance, diseases common in later life, including diabetes, hypertension, and stroke, can impair the blood-brain barrier that normally blocks many contaminants from reaching the sensitive tissues of the brain.
All these factors can result in older adults being particularly susceptible to health effects from industrial chemicals. EPA scientists summarize research showing these links between environmental exposures and disease in the elderly, including neurotoxicity, cancer, cardiovascular and pulmonary morbidity, and inflammatory responses (Geller and Zenick 2005). Increasingly, scientists are also questioning the role of industrial pollutants in a range of degenerative diseases of aging, including Parkinson's and Alzheimer's disease.
About the Participants
Four mothers and their daughters participated in "Across Generations," donating blood and urine for the study in November and December 2005. Currently living in New York, Colorado, Washington DC, and California, these women range in age from 17 to 62. On average, more than 90 percent of the chemicals found in the daughters were also found in their mothers. These mothers and daughters, and the common ground they find in their body burden of chemicals, are described below. Based on each daughter's age, we provide estimates of how much of her inherited load of industrial pollution remains in her body. These remaining, inherited loads of pollution now likely represent just one portion of each daughter's total body burden of industrial chemicals, which for these four women has continued to accumulate over the two to nearly three decades that have passed since their births.

Annette Gellert and her daughter Heather Gellert live in northern California. Annette is a business executive, and Heather, at age 17, is a high school student. Of the 34 chemicals found in Heather's body, 30 (88 percent) were also found in her mother. According to measurements of chemical persistence in the body, Heather still carries an estimated 27 percent of the Scotgard (PFOS), 7 percent of the Teflon (PFOA), and 62 percent of the lead passed on to her from her mother, across the placenta and through breast milk.

Ann Hunter-Welborn, a community activist in Encinitas, California, and her daughter Jessica Welborn, a PhD student from San Francisco, share all but two of the 29 industrial chemicals found in Jessica's blood and urine. Jessica still harbors in her body nearly half of the lead passed on to her from her mother across the placenta and through breast milk, and an estimated 12 percent of the Scotchgard (PFOS) and one percent of the Teflon (PFOA) she inherited from her mother at birth.
Martha Davis, an environmental scientist from Denver, Colorado, and her daughter Emily Sayrs, a student, claim the highest shared burden of industrial chemicals of any mother and daughter pair tested. All but one of the 41 chemicals in Emily's blood and urine were also found in her mother. Emily continues to bear an estimated 62, 27, and 7 percent, respectively, of the lead, Scotchgard, and Teflon passed on to her from her mother at birth.

Adelaide Gomer is vice president of a charitable foundation in New York, and her daughter Alicia Wittink is a a Washington, D.C.-based consultant. More than 92 percent of the 26 chemicals found in Alicia were also present in her mother. When Alicia gave birth to her daughter in fall 2005, she still carried in her body an estimated 47, 12, and one percent of the lead, Scotchgard, and Teflon she inherited from her mother — through her placenta and breast milk she passed down some of this same pollution to her own daughter.
About the Chemicals
In this study two major laboratories tested blood and urine from 22 people, including four mothers and their daughters, for 70 common consumer product chemicals, including plasticizers known as phthalates, perfluorochemicals (PFCs) in the family of Teflon and Scotchgard chemicals, brominated flame retardants called PBDEs, lead, and methylmercury. Below is information on exposure sources, safety standards, and health concerns for each of these chemicals or chemical families.
![]() Teflon chemicals (Perfluorochemicals, or PFCs). PFCs are ubiquitous stain- and grease-proof coatings on furniture, carpet, clothing and food packaging. They include Teflon, Scotchgard, Gore-Tex, and coatings on microwave popcorn bags, butter boxes, and fast food containers. They never break down in the environment, and can build up in the body over time. Some chemicals in this family move through the atmosphere easily, and contaminate wildlife globally, from polar bears to cormorants. In laboratory studies they have been linked to multiple types of cancer (breast, liver, testicular, and pancreas) and birth defects. In studies of workers they are associated with prostate cancer and elevated risk of stroke. Safe levels of exposure have not yet been set.
Teflon chemicals (Perfluorochemicals, or PFCs). PFCs are ubiquitous stain- and grease-proof coatings on furniture, carpet, clothing and food packaging. They include Teflon, Scotchgard, Gore-Tex, and coatings on microwave popcorn bags, butter boxes, and fast food containers. They never break down in the environment, and can build up in the body over time. Some chemicals in this family move through the atmosphere easily, and contaminate wildlife globally, from polar bears to cormorants. In laboratory studies they have been linked to multiple types of cancer (breast, liver, testicular, and pancreas) and birth defects. In studies of workers they are associated with prostate cancer and elevated risk of stroke. Safe levels of exposure have not yet been set.
The level of total PFCs (the sum of 13 individual chemicals) in serum, averaged 1.5 times higher in mothers than their daughters, and ranged from 6.7 to 42.5 parts per billion (nanograms per milliliter of serum).
One PFC chemical accounted for an average of 62 percent of the total PFC exposures among mothers and daughters: 3M's Scotchgard chemical (PFOS), which the company used from the 1950's until its phaseout over health concerns in 2000 (Scotchgard has since been reformulated). The PFOS in women and girls from this study is likely from past exposures.
Most PFCs are difficult to excrete, and remain in the body for a long time. The body burden levels in this study reflect exposures over a period ranging from decades to months prior to blood sampling, depending on the particular PFC chemical.
PFC manufacturers are facing a barrage of legal and regulatory pressure over the toxicity of these compounds and their near universal presence in human blood. The Environmental Protection Agency (EPA) forced 3M to take Scotchgard off the market in 2000 when company tests revealed widespread human blood pollution and birth defects in laboratory studies. 3M has since reformulated the product. Under pressure from EPA, in February 2006 DuPont, 3M and other major manufacturers agreed to dramatically reduce their environmental pollution for PFCs. For at least $120 million DuPont recently settled two cases over drinking water pollution and the company's suppression of data on birth defects and human blood pollution. Two chemicals in the PFC family and in the blood of all eight mothers and daughters tested are proposed for global phase-out under the terms of the Persistent Organic Pollutants (POPs) treaty known as the Stockholm Convention. These are PFOS (pre-2000 Scotchgard) and PFOA, a Teflon manufacturing aid and breakdown product of PFC coatings that is linked to birth defects and cancer.
EPA is currently conducting an assessment to estimate health risks from consumers' exposures to PFCs, but in the meantime, the chemicals are still the standard in stain- and grease-proof coatings in everything from blue jeans to French fry boxes.
To minimize exposures to PFCs and their impacts on the environment, we recommend that consumers phase out their use of Teflon and other non-stick pans; decline optional stain-proof coatings on furniture and carpet; avoid buying clothing advertised as stain-proof; and eat fewer greasy, pre-packaged foods, which tend to be held in containers coated with PFCs.
![]() Mercury. In blood samples we tested for methylmercury, the form of mercury associated with seafood contamination, not dental fillings or vaccine preservatives. In general, levels reflect the contamination in seafood eaten over the several months prior to blood sampling.
Mercury. In blood samples we tested for methylmercury, the form of mercury associated with seafood contamination, not dental fillings or vaccine preservatives. In general, levels reflect the contamination in seafood eaten over the several months prior to blood sampling.
The level of methylmercury averaged 5.3 times higher in mothers than their daughters, and ranged from 0.9 to 9.8 parts per billion (nanograms per gram of serum). For 3 mothers, methylmercury levels were higher than the federal safety standard of 5.8 ppb set for pregnant or nursing women and young children to protect against brain damage. While the government has not yet set a safe level to protect non-pregnant adults, the National Academy of Sciences found that mercury-driven risks for immune disorders and cardiovascular disease may occur at even lower levels than those associated with brain impairment.
People can lower their mercury exposures by changing the types of seafood they eat. Body burden levels will decline over time: on average, the human body can excrete half of its mercury load over a period of about two months. We recommend avoiding fish high in mercury, including swordfish, red snapper, tuna, and halibut, and eating fish known to be low in mercury.
![]() Bisphenol A (BPA) and Bisphenol A diglycidyl ether metabolite (BADGE-40H). BPA is used to make polycarbonate plastics, and is also used to make a chemical called BADGE that forms epoxy resins lining metal food cans. We tested blood for BPA as well as BADGE, in the form in which it is found in the body (BADGE-40H). Recent studies have linked BPA to many adverse health effects in mice and rats at doses that overlap typical human exposures. These effects include early puberty, breast cancer, diabetes, and low sperm count. Safe levels of exposure for BPA and BADGE have not been set.
Bisphenol A (BPA) and Bisphenol A diglycidyl ether metabolite (BADGE-40H). BPA is used to make polycarbonate plastics, and is also used to make a chemical called BADGE that forms epoxy resins lining metal food cans. We tested blood for BPA as well as BADGE, in the form in which it is found in the body (BADGE-40H). Recent studies have linked BPA to many adverse health effects in mice and rats at doses that overlap typical human exposures. These effects include early puberty, breast cancer, diabetes, and low sperm count. Safe levels of exposure for BPA and BADGE have not been set.
BPA was detected in the blood of 1 daughter in this study, at 2.6 parts per billion (nanograms per milliliter of serum). BADGE-40H was detected in 3 mothers and 3 daughters, at levels ranging from 0 to 174 parts per billion (nanograms per milliliter of serum). National exposure levels are not available: the Centers for Disease Control and Prevention is expected to publish the first national study of BPA body burden levels in 2007.
BPA is currently under intense scientific and regulatory debate because of concerns that it might pose risks to human reproduction. A major government safety panel is due to convene on the subject in the fall of 2006. Seven prior government reviews outside the U.S. have deemed current BPA exposures within safe limits, but none has assessed the reproductive risks demonstrated in what are now more than 100 low-dose studies.
To reduce exposures to BPA and its epoxy resin (BADGE), we recommend avoiding drinking water from hard, transparent plastic bottles (including water cooler bottles, and Nalgene), and avoiding regularly consuming foods from metal cans.
![]() Lead. Lead was found in the blood of all mothers and daughters tested, at levels below the current action level of 10 ug/dL, set to protect children against the well-established brain damage associated with this neurotoxic metal. While an action level has not been set for adults, lead is thought to pose risks at any level, and scientists are beginning to question the role that a lifetime of lead exposure may play in degenerative diseases of aging.
Lead. Lead was found in the blood of all mothers and daughters tested, at levels below the current action level of 10 ug/dL, set to protect children against the well-established brain damage associated with this neurotoxic metal. While an action level has not been set for adults, lead is thought to pose risks at any level, and scientists are beginning to question the role that a lifetime of lead exposure may play in degenerative diseases of aging.
The level of lead in blood averaged 2.1 times higher in mothers than their daughters, and ranged from 5 to 36 parts per billion (nanograms per milliliter of serum). Mothers' higher levels were expected, since lead builds up in the body over time.
In general, Americans' lead exposures are much lower now than they were before about 1980, when the use of leaded gasoline and lead-based house paint was restricted. Now, most people are exposed to lead through their drinking water (it leaches from lead pipes, pipe solder, and brass fixtures in the home) or through dust from chipping paint in older homes.
Lead stays in the body for a long time: on average people excrete half of any lead exposure over a period of 30 years. The levels measured in blood reflect both childhood exposures and more recent exposures. While it is not possible to significantly reduce your current body burden of lead, it is a good idea to minimize future exposures.
We recommend that people test their tap water for lead with a simple home test kit and install a water filter if needed. Since lead can accumulate in water pipes overnight, we recommend that running the tap water for at least 60 seconds, until it runs cold, before drinking it. And we recommend keeping paint in older homes in good repair, having chipped surfaces repaired using wet sanding methods to minimize dust formation.
![]() Phthalates. Phthalates were found in all eight mothers and daughters tested. These chemicals are common plastic softeners and solvents in a wide variety of consumer products, including cosmetics, paint, food packaging, and plastics including children's toys. In many laboratory studies phthalates are linked to birth defects of the male reproductive system, including a penis deformity called hypospadias, and undescended testicles, a condition linked to infertility and testicular cancer. Studies of phthalates in people have been few, but tentatively link the chemicals to impaired sperm motility, demasculinization of baby boys, asthma, and alteration of reproductive hormone levels in breast-fed boys.
Phthalates. Phthalates were found in all eight mothers and daughters tested. These chemicals are common plastic softeners and solvents in a wide variety of consumer products, including cosmetics, paint, food packaging, and plastics including children's toys. In many laboratory studies phthalates are linked to birth defects of the male reproductive system, including a penis deformity called hypospadias, and undescended testicles, a condition linked to infertility and testicular cancer. Studies of phthalates in people have been few, but tentatively link the chemicals to impaired sperm motility, demasculinization of baby boys, asthma, and alteration of reproductive hormone levels in breast-fed boys.
In general, people excrete phthalates from the body fairly rapidly, so body burden levels reflect exposures in the day or two prior to sampling. Because of their ubiquity in consumer products and the human body, phthalates are the subject of much recent scientific and regulatory action and debate. Of the seven phthalates found in this study, the EU has banned DEHP from children's toys, and DBP and DEHP from cosmetics. None are banned in the U.S., but some manufacturers have removed them from toys and personal care products because of public pressure.
To reduce exposures to phthalates, we recommend avoiding nail care products containing "dibutyl phthalate" (read the ingredient label); avoiding products with "fragrance" noted on the label, which commonly contains the phthalate DEP; avoiding cooking in plastic; buying a non-plastic shower curtain; and using paints and other hobby products in well-ventilated areas. Also, don't let children chew on soft plastic toys.
![]() Brominated flame retardants (PBDEs). PBDEs, or polybrominated diphenyl ethers, are brominated flame retardants used in foam furniture and plastics for electronics like televisions and computers for the past 25 years. These chemicals interfere with the thyroid gland, which controls metabolism and growth. They may jeopardize brain development in children, and they raise concerns with respect to thyroid disease in adults. Safe exposure levels have not yet been established.
Brominated flame retardants (PBDEs). PBDEs, or polybrominated diphenyl ethers, are brominated flame retardants used in foam furniture and plastics for electronics like televisions and computers for the past 25 years. These chemicals interfere with the thyroid gland, which controls metabolism and growth. They may jeopardize brain development in children, and they raise concerns with respect to thyroid disease in adults. Safe exposure levels have not yet been established.
The level of total PBDEs (the sum of all individual chemicals) in serum averaged 1.6 times higher in mothers than their daughters, and ranged from 9.4 to 41 parts per billion (nanograms per milliliter of serum). National exposure levels are not available; the Centers for Disease Control and Prevention is expected to publish the first national study of PBDE body burden levels in 2007.
Many PBDEs are difficult to excrete, and remain in the body for a long time. Your body burden levels reflect your exposures over a period ranging from years to weeks prior to your blood draw, depending on the particular PBDE chemical.
In 2005 manufacturers stopped selling two of the most toxic PBDE commercial mixtures because of concerns over its ubiquity in human blood and breast milk: "Penta," (predominantly containing chemicals called PBDE-99 and PBDE-47 chemicals) and "Octa," (predominantly comprising PBDE-183). These same mixtures were banned in Europe two years earlier, although European manufacturers had never used them widely. Penta and Octa chemicals remain in many U.S. homes, primarily in foam furniture, and they commonly contaminate house dust.
Penta and Octa chemicals may continue to build up in people's bodies even after their phase-out: laboratory data shows that the common "Deca" form of PBDEs (PBDE-209) used in electronics, including computer and television casings, may break down into Penta and Octa in the environment and the body. Many states are considering banning Deca as well as Penta and Octa formulations.
Despite the potential health risks and the ability of these chemicals to accumulate in the human body, the Consumer Product Safety Commission has issued national fire-proof mattress standards effective July 2007 that could increase the use of flame retardants. Developed primarily to protect against cigarette fires, these standards would result in the widespread use of brominated flame retardants in mattresses. Because such standards have long been in place in California, several companies offer mattresses free of flame retardants as an alternative for concerned consumers.
Acknowledgments
About This Report
We would like to thank Rachel's Network for their support of and participation in this study. Rachel's Network is a 501(c)(3) nonprofit organization dedicated to creating a network of women conservation funders to be an influential voice for the environment.
This study was made possible by grants from members of Rachel's Network, the The Beldon Fund, The John Merck Fund, The Nathan Cummings Foundation, and The Johnson Family Foundation. The opinions expressed in this report are those of the authors and do not necessarily reflect the views of the supporters listed above. EWG is responsible for any errors of fact or interpretation contained in this report.
This study was designed and implemented by Renee Sharp, Sean Gray, and Jane Houlihan of the Environmental Working Group. The report was authored by Jane Houlihan, and designed by T.C. Greenleaf and Carrie Gouldin.
EWG is a nonprofit research organization with offices in Washington, DC and Oakland, CA. EWG uses the power of information to educate the public and decision-makers about a wide range of environmental issues, especially those affecting public health.


