
DuPont’s Dirty History
In 2005 the Environmental Protection Agency fined chemical giant DuPont a record $16.5 million over its decades-long cover-up of the health hazards of C8, also known as PFOA. One of a family of perfluorinated chemicals, or PFCs, PFOA was a key ingredient in making Teflon, the non-stick, waterproof, stain-resistant “miracle of modern chemistry” used in thousands of household products.
DuPont had long known that PFOA caused cancer, had poisoned drinking water in the mid-Ohio River Valley and polluted the blood of people and animals worldwide. But it never told its workers, local officials and residents, state regulators or the EPA. Eventually, research by federal officials, EWG and other public interest groups found that the blood of almost all Americans was contaminated with PFCs. And in 2006 the EPA confirmed that PFOA is a probable human carcinogen.
Ten years later, a new EWG investigation shows that Americans are still threatened by PFCs and that the victims of DuPont’s heinous actions are still awaiting justice. Click here to read EWG’s full report.
PFCs in your life
PFOA is no longer in used in the United States by the end of 2015, but DuPont and other companies are using related chemicals that may not be much safer. These next-generation PFCs are used in greaseproof food wrappers, waterproof clothing and other products. Few have been adequately studied for safety, and chemical companies hide critical information about most of them as trade secrets.
EWG wants to make sure that all consumers know about these new chemicals. We have created the resources you need to make smarter, healthier choices, including a brand new consumer guide. Download EWG’s consumer guide, complete with tips and tricks to keep PFCs out of your home:
Tell DuPont to keep its promises
It’s been 10 years since the EPA’s history-making judgment against DuPont and PFOA, but its victims are still seeking justice. DuPont has failed to clean up water supplies, is shirking its promise to monitor the health of the communities it poisoned and is gearing up to fight in court against paying damages to its victims. EWG – and the victims of DuPont – need you to take action today!
Additional Resources
EWG's Chemical Industry Archives - See the documents that show how DuPont hid PFC pollution for decades.
The Madrid Statement - A scientific consensus statement regarding the persistence and potential for harm of PFCs.
Green Science Policy's Consumer Guide to Highly Fluorinated Chemicals.
Dupont Keep Your Promises Campaign
Executive Summary
 In 2005, the U.S. Environmental Protection Agency fined chemical giant DuPont $16.5 million over its decades-long cover-up of the health hazards of a substance known as C8. One of a family of perfluorinated chemicals, or PFCs[1], C8 was a key ingredient in making Teflon, the non-stick, waterproof, stain-resistant “miracle of modern chemistry” used in thousands of household products.
In 2005, the U.S. Environmental Protection Agency fined chemical giant DuPont $16.5 million over its decades-long cover-up of the health hazards of a substance known as C8. One of a family of perfluorinated chemicals, or PFCs[1], C8 was a key ingredient in making Teflon, the non-stick, waterproof, stain-resistant “miracle of modern chemistry” used in thousands of household products.
Internal documents revealed DuPont had long known that C8, also known as PFOA, caused cancer, had poisoned drinking water in the mid-Ohio River Valley and polluted the blood of people and animals worldwide. But the company never told its workers, local officials and residents, state regulators or the EPA. After the truth came out, research by federal officials and public interest groups, including EWG, found that the blood of almost all Americans was contaminated with PFCs, which passed readily from mothers to unborn babies in the womb. In 2006 the EPA confirmed that PFOA is a likely human carcinogen.
The 2005 fine against DuPont remains the largest ever levied by the EPA. DuPont did not admit guilt but promised to phase out production and use of C8/PFOA by this year – 2015. Also in 2005, DuPont entered into a settlement valued at well over $300 million in a class-action lawsuit brought on behalf of approximately 70,000 people living near its Washington Works plant in Parkersburg, W. Va., where it had long made and used C8 and dumped the waste in waterways and landfills. Under the terms of the settlement, DuPont promised to clean up water supplies, fund a panel of scientists to determine what diseases C8 caused and pay to monitor the health of affected residents for the rest of their lives.
The 2005 fine, settlement and phase-out were widely hailed as a public health victory and justice for the victims. But 10 years later, a new EWG investigation shows that it remains uncertain whether Americans are safe from the threat of PFCs and whether justice will be done for the victims.
Production, use and importation of PFOA has ended in the United States, but in its place DuPont and other companies are using similar compounds that may not be much – if at all – safer. These next-generation PFCs are used in greaseproof food wrappers, waterproof clothing and other products. Few have been tested for safety, and the names, composition and health effects of most are hidden as trade secrets. With the new PFCs’ potential for harm, continued global production, the chemicals’ persistence in the environment and presence in drinking water in at least 29 states, we’re a long way from the day when PFCs will be no cause for concern.
In a just-published paper, 14 international scientists have sounded the alarm, calling for tighter controls on all PFCs lest the tragic history of C8 repeat itself. Writing in Environmental Health Perspectives, they likened the new PFCs (which they refer to as PFASs[2]) to the chemicals that replaced another group of fluorine-based substances found in the 1980s to be depleting Earth’s protective ozone layer. Although those chemicals were banned worldwide under a 1987 treaty, the scientists wrote, the alternatives are also harmful:
Global action through the Montreal Protocol successfully reduced the use of the highly persistent ozone-depleting chlorofluorocarbons (CFCs), thus allowing for the recovery of the ozone layer. However, many of the organofluorine replacements for CFCs are still of concern due to their high global warming potential. It is essential to learn from such past efforts and take measures at the international level to reduce the use of PFASs in products and prevent their replacement with fluorinated alternatives in order to avoid long-term harm to human health and the environment. (Blum et al 2014)
Even as the threat from the new generation of PFCs grows, DuPont is trying to skirt the consequences of its toxic irresponsibility.
- The company has ducked a commitment to treat the water supply in Parkersburg, the largest affected water system in the mid-Ohio Valley, on the grounds that levels of C8 in the water were originally lower – by a tiny amount – than the 2005 settlement’s cleanup threshold. More recent tests found the chemical at levels above the threshold. (Jeffersonian 2006) DuPont has also fought the cleanup claims of another water district across the river in Little Hocking, Ohio. (U.S. District Court 2015a)
- DuPont forced out a trusted local health services company that was initially hired to run the medical monitoring program in West Virginia and Ohio, replacing it with a New York law firm notorious for helping corporate polluters lowball their liability payouts. Invoices released by a local citizens’ group show the firm was paid $9 million through January 2015 but has paid out only about $50,000 to residents. (Saulton 2015)
- In July 2015, DuPont will spin off its Specialty Chemicals unit, which made C8/PFOA and now makes the replacement chemicals for Teflon and other products, to a new corporation called Chemours. Securities and Exchange Commission filings indicate that this may transfer DuPont’s legal liability for damage from C8 to Chemours. This could shield DuPont from full liability and allow the smaller company to claim that it has insufficient assets to pay compensation for the damage done in the mid-Ohio Valley and other places where C8 was made or used. (U.S. District Court 2015b)
- In September 2015, a trial is scheduled in U.S. District Court in Columbus, Ohio, consolidating personal injury claims against DuPont by more than 2,500 residents of the mid-Ohio Valley. In pre-trial maneuvering, DuPont tried to renege on a key promise it made in the 2005 settlement: that in the case of any resident who drank contaminated water and sued over a disease the science panel determined has a probable link to C8 exposure, DuPont would concede that C8 could cause the disease in that group of people. The trial judge ruled against DuPont, but the gambit showed that the company is still seeking to shortchange its victims. (U.S. District Court 2014)
Even as DuPont maneuvers to minimize its responsibility for letting a known hazardous compound contaminate the homes, water and bodies of all Americans, the public remains vulnerable to future disasters because of the gaping holes in the nation’s chemical safety net.
Under the broken 1976 Toxic Substances Control Act, or TSCA, EPA has managed to limit or ban only five dangerous chemicals over nearly 40 years. (GAO 2013) The lack of teeth in the law allowed DuPont to phase out C8 over 10 years while the company continued to reap profits and prevented EPA from punishing DuPont more severely. Meanwhile the law has allowed DuPont and other companies to rush next-generation PFCs to market without first proving they’re safe.
Now the American Chemistry Council, the lobbying arm of the chemical industry, and its allies in Congress are pushing a sham “reform” bill to replace the Toxic Substances Control Act. The industry-friendly “reforms” in the legislation drafted by the trade group (McCumber 2015) would continue to hobble EPA’s ability to protect the public from untested and unsafe chemicals and prevent states from taking more protective action on their own.
None of this is acceptable. DuPont must be held to its promises to clean up the mid-Ohio Valley and compensate those who were harmed. The EPA and governments worldwide must act swiftly to thoroughly assess and control the hazards of next-generation PFCs. Most importantly, Congress must learn from the tragedy of C8 and enact an effective chemical safety law that protects public health, not the industry’s profits.
[1] Poly- and perfluoroalkyl substances. Among scientists this more precise term and acronym are commonly used, but this report will use ‘PFCs,” as the chemicals have been known for most of their history.
[2] The term PFCs refers to both per- and polyfluorinated chemicals in which all or many of the hydrogen atoms have been replaced by fluorine. In the environment, polyfluorinated chemicals can break down to perfluorinated chemicals.
From Lab Accident to Global Pollutant
In 1938, Roy J. Plunkett, a chemist at E. I. du Pont de Nemours and Company, accidentally invented “the most slippery substance on Earth” – PTFE,[1] the first compound in the family of perfluorinated chemicals, or PFCs, to be marketed commercially. (Lyons 2007) Patented as Teflon, it was ultimately used in more than 3,000 products – nonstick cookware, waterproof clothing, camping gear, even dental floss. At their peak, Teflon sales reached $1 billion a year. (Haber 2005) In 2008, the United States alone used 100 million pounds of PTFE, about one-fourth the worldwide total. (EPA 2009)
An essential ingredient in making Teflon was another PFC known as PFOA.[2] PFOA is often called C8 after the number of carbon atoms in its chemical chain. PFCs are made up of chains of carbon atoms of varying lengths that bond strongly to fluorine atoms, yielding chemicals that do not break down in the environment and take years to decades to pass from our bodies. (Steenland 2010) (Perfluorinated chemicals with eight or more carbon atoms are called “long-chain” PFCs; the replacements now hitting the market have shorter fluorinated carbon chains.)
C8/PFOA itself was not in finished products but was used to make Teflon and was a byproduct of the process. Until 2000, the major U.S. manufacturer of C8 was 3M Company, which also made a closely related compound called PFOS.[3] [4] PFOS was a key ingredient in 3M’s stain-resistant Scotchgard, which had thousands of consumer and industrial applications from fabric sprays to fast food wrappers.
Even as Teflon and Scotchgard were earning big profits for DuPont and 3M, the companies were accumulating evidence that PFOA and PFOS were hazardous. For decades the companies kept the increasingly alarming information secret. (EWG 2002A) The truth only emerged after EPA forced 3M to phase out PFOS and lawsuits were brought against DuPont for polluting of mid-Ohio Valley water supplies with C8/PFOA. Table 1 summarizes some of these secret studies.
Table 1. Decades of Secret Studies by DuPont and 3M
|
1961 |
A DuPont toxicologist warns that Teflon chemicals cause liver enlargement in rats and rabbits. |
|
1962 |
DuPont scientists have 40 volunteers smoke cigarettes laced with Teflon. Ninety percent of the most highly exposed group develop flu-like symptoms known as polymer fume fever. |
|
1973 |
DuPont finds there is no safe level of exposure to C8/PFOA in animals. |
|
1976 |
3M begins testing some workers’ blood for PFOA and finds it in almost every one tested. |
|
1978 |
3M finds that PFOA is “completely resistant” to breakdown in the environment. |
|
1979 |
3M finds PFOS in the blood of five workers in Alabama. Fish in the Tennessee River, where up to 1 million pounds of PFOS waste were dumped each year, are found to have significant concentrations of the chemical in their blood, evidence of bioaccumulation. |
|
1981 |
3M finds that PFOA causes birth defects in rats. |
|
1981 |
DuPont finds PFOA in umbilical cord blood from one baby and blood from a second baby born to female workers at the Washington Works plant. |
|
1981 |
Two of seven children of women working at Washington Works are found to have birth defects of the eye, tear duct or nose. DuPont transfers “all potentially exposed female employees” out of the plant but does not tell them why. |
|
1983 |
3M doctors warn that organic fluorine levels in workers’ blood are steadily rising, evidence that PFCs accumulate faster than the body can eliminate them. |
|
1984 |
DuPont finds PFOA in tap water in two mid-Ohio Valley communities. Tests continue for 17 years before DuPont informs any area water suppliers. |
|
1992 |
Data on workers at DuPont’s Washington Works plant reveal an excess of deaths from cancer and leukemia. |
|
1993 |
A 3M study of employees at a PFOA plant finds twice as many deaths from prostate cancer as in the general population. |
|
1997 |
3M looks worldwide for clean blood samples to compare to its workers’ blood but finds only one source not contaminated with PFOS – preserved blood of soldiers who died in the Korean War, before Scotchgard products spread worldwide. |
|
1998 |
3M finds that PFOS causes liver cancer in rats. Despite federal law prohibiting the use in food of any substance that causes cancer in animals, 3M continued until 2000 to petition the FDA to allow PFOS in microwave popcorn bags. |
|
1999 |
3M study in six East Coast cities finds PFCs in supermarket food, rivers and lakes, drinking water sources and tap water. |
Source: Environmental Working Group, from DuPont and 3M documents in EWG’s Chemical Industry Archives, www.chemicalindustryarchives.org.
The discovery of PFOS in blood samples worldwide in 1997 must have spooked 3M. It started submitting selected studies to the EPA while arguing for the right to continue using the chemical in some applications even as it reformulated Scotchgard and related products. Documents in EPA’s files made public by EWG don’t give details of the agency’s negotiations with 3M, but the end came quickly after April 2000, 3M submitted a study showing deaths among monkeys exposed to low levels of PFOS. (EWG 2002B)
The next month 3M issued a vague one-page press release saying it would end production and use of PFOS by the end of 2002. Continuing to hide the truth, 3M cited concerns over what it said was new information that the chemical had been “detected broadly at extremely low levels in the environment and people. All existing scientific knowledge indicates that the presence of these materials at these very low levels does not pose a human health or environmental risk.” (3M 2000) Two days later, The New York Times reported that EPA had forced 3M’s hand: “Agency officials said that if 3M had not acted they would have taken steps to remove the product from the market.” (Barboza 2000)
As 3M phased out PFOS, it also stopped making PFOA, but DuPont started making its own at a Fayetteville, N.C., plant. Since 1951 it had been using the chemical at the Washington Works plant and disposing of it in area waterways, landfills and unlined pits, as well as polluting the air with it through the plant’s smokestacks.
In 1984, secret tests by DuPont found C8/PFOA in the drinking water of two nearby communities on either side of the Ohio River. An internal DuPont memo recommended elimination of “all C8 emissions at our manufacturing sites in a way … which does not economically penalize the business.” Instead, DuPont chose to significantly increase production and keep quiet about the water pollution and health hazards (Lyons 2007)
Then cattle started dying on the Tennant family farm.
In the early 1980s the Tennants had sold some land south of the Washington Works plant to DuPont for what they thought would be a non-hazardous landfill. A creek ran through the landfill and into the Tennants’ pasture, and the water soon ran black and bubbly. By 1998 they had lost several hundred cows. They hired Robert Bilott, a Cincinnati attorney with roots in the area, to sue DuPont.
In a DuPont document Bilott found a reference to a chemical he knew nothing of: PFOA. He went to court to get more documents, which revealed the secret water tests. The Tennants settled out of court and the terms were sealed. But with the knowledge that public water supplies had been contaminated, in 2001 Bilott brought a class-action suit against DuPont on behalf of more than 50,000 area residents. (Lyons 2007)
As he pursued the case, Bilott obtained tens of thousands of damning documents. He alerted local and state authorities but got little response. He wrote to the EPA, detailing numerous violations of the Safe Drinking Water Act, the Toxic Substances Control Act and other laws. EPA did nothing. EWG then obtained many of the same documents from public court files, published the documents online in a series of reports that brought the case to national attention and pressured EPA to act. In 2003 the agency opened an emergency review of PFOA, including possible regulatory action under the Toxic Substances Control Act.
Over the next three years, the secret studies continued to surface, but each time DuPont insisted that PFOA posed no health threat. But Robert Bilott, EWG and others kept the pressure on EPA to act. After intense negotiations that included the prospect of criminal charges by the Justice Department, DuPont agreed in 2006 to “voluntarily” phase out the chemical by 2015. EPA assessed a record $16.5 million fine against the company for not disclosing the health studies on PFOA as required by the Toxic Substances Control Act, although DuPont maintained that it had not broken the law. And in 2006 EPA’s Science Advisory Panel classified PFOA as “likely to be carcinogenic to humans.” (EPA 2006)
In 2005, DuPont settled the class-action suit by the residents under a unique agreement, with provisions not previously seen in chemical pollution cases. DuPont paid $70 million in damages upfront and promised to pay for a state-of-the-art cleanup of the Parkersburg area’s water supplies. DuPont also agreed to fund an independent panel of scientists – the C8 Science Panel – to study the links between C8 and various diseases. Under the settlement, if the science panel found probable links between C8 exposure to residents and disease, those residents could pursue damage claims against DuPont. If such links were found, DuPont would pay up to $235 million to provide medical monitoring for the exposed residents.
[1] Polytetrafluoroethylene.
[2] Perfluorooctanoic acid.
[3] Perfluorooctanesulfonic acid.
[4] The Appendix provides a fuller explanation of the chemical structure of PFOA, PFOS and other PFCs.
PFC Contamination: No Place to Hide
PFCs in people
PFCs were first detected in human blood in 1976. Dr. Donald Taves of the University of Rochester's School of Medicine and Dentistry found that some of the fluoride in his own blood was organic and not related to the fluoride added to public drinking water supplies. Taves and his collaborators tentatively identified one of the chemicals as PFOA. (More than 20 years later, 3M said this was likely a misidentification of PFOS.) (Taves 1976, 3M 1999)
Over the next two decades, more than a dozen studies were published on blood levels of PFCs in the general population. In 2001 – four years after its futile worldwide search for blood samples not contaminated by PFOA and a year after it agreed to phase out PFOA and PFOS – 3M submitted a study to the EPA that found PFOA in the blood of 96 percent of 598 children tested in 23 states and the District of Columbia. In 2003-2004, the National Health and Nutrition Examination Survey, conducted by the Centers for Disease Control, found PFOA, PFOS and 10 other long-chain PFCs in 98 percent of a representative sample of the U.S. population. (NHANES 2015)
In 2005, EWG and Commonweal, a public health non-profit, obtained through the Red Cross samples of umbilical cord blood of 10 American newborns. Testing by two independent laboratories found that all ten had PFOA, PFOS or seven other long-chain PFCs in their blood – the first publicly reported tests to confirm that these chemicals could be passed from mothers to babies in the womb. (EWG 2005) In 2009, EWG and Rachel’s Network, another non-profit, commissioned tests on the cord blood of 10 newborns from African-American, Hispanic and Asian-American mothers and again found PFCs, including some of the next-generation replacements, in every one. (EWG 2009)
After decades of pollution from DuPont’s Washington Works plant in the mid-Ohio Valley, levels of C8/PFOA in people there were extraordinarily high. In 2003-2004, the National Health and Nutrition Examination Survey had found that the average level of PFOA in Americans was 4 parts per billion. (NHANES 2007) The C8 Science Panel found that the average in blood samples from the mid-Ohio Valley was 83 parts per billion. (Steenland 2009) The median level – the point at which half the samples were above and half below – was 28 parts per billion.
Blood levels in some groups sampled near the plant were even higher. The median level of C8 in people living closest to the plant, whose drinking water that came from the Little Hocking, Ohio, water district, was more than 224 parts per billion. The median level in workers currently employed at the plant was more than 147 parts per billion, and in former workers it was almost 74 parts per billion. C8 levels were higher in children, in people who ate local vegetables and in those who drank well water rather than public water. (Steenland 2009)
How do PFCs get into us?
People get contaminated with PFCs in just about every way imaginable – from drinking water, food and food packaging, indoor air, household and workplace air, carpet and furniture treatments, clothing, cosmetics, non-stick cookware and many other products. There is much uncertainty about how much exposure comes from each source. Early studies pointed to food and water as major sources and suggested that consumer goods were not significant. (Trudel 2008, Washburn 2005) A later study of blood serum levels found that indoor air in the workplace could be an important exposure pathway. (Fraser 2011)
Recently, EPA has suggested that the widespread presence of PFCs in human blood indicates that products treated to be stain-proof or waterproof could be a source. (EPA 2015B) In occupants of homes that are regularly treated with stain-resistant sprays, PFC levels can be much higher than in the general public. In children, who come into closer and more frequent contact with carpets and dust, levels are almost always higher than in adults. (Beesoon 2010, Mondal 2012, Toms 2009)
In 2009, EPA measured the chemical content of 116 products commonly treated with PFCs and estimated the typical amounts in a U.S. home. (Guo 2009) The results showed that more than 95 percent of PFCs in the home came from carpets and carpet treatments.
Upholstery, floor treatments and textiles also bring PFCs into the home. Non-stick pans contribute relatively small amounts. (Figure 1) In some people the indoor environment accounted for half of typical PFOA and PFOS exposure, but for the general population, food accounted for 67-to-84 percent of PFOA exposure and 88-to-99 percent of PFOS exposure. (Haug 2011)
Figure 1. Sources of PFC exposure in a typical U.S. home.
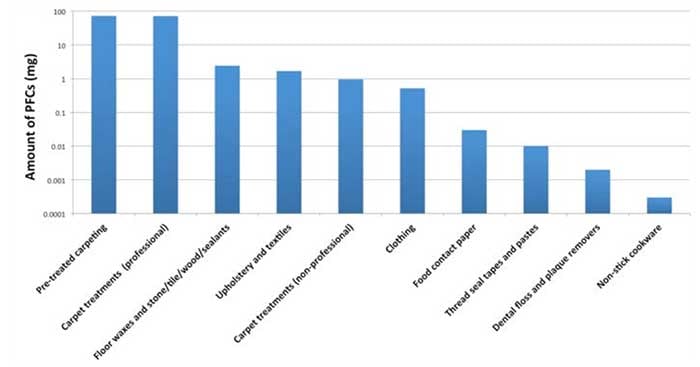
Source: Environmental Working Group, from EPA. http://www.oecd.org/env/48125746.pdf
How long do PFCs remain in the body?
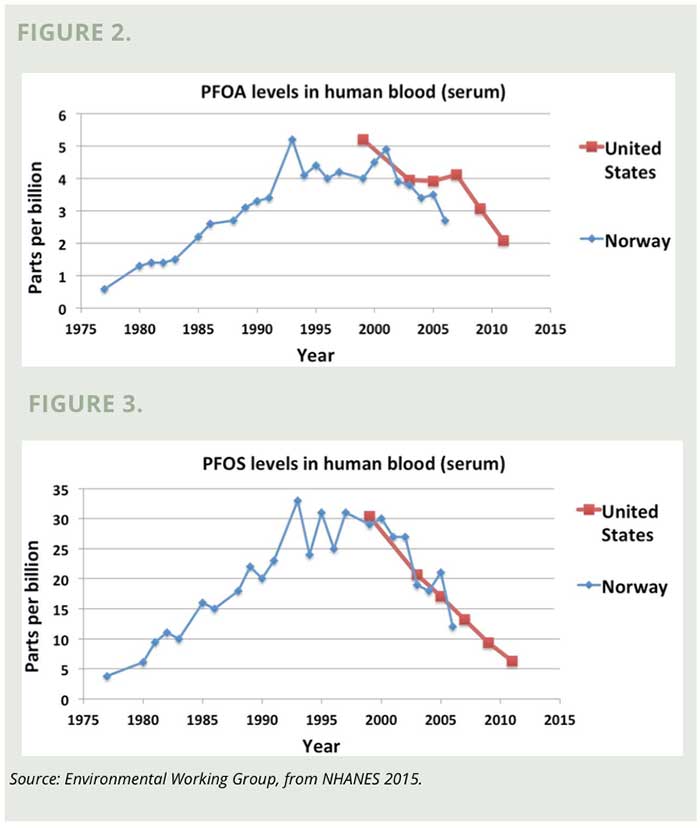
The National Health and Nutrition Examination Survey (NHANES) has been testing for PFCs in the general population since the late 1990s and measuring the chemicals’ half-lives. (The half-life is how long it would take for half of the chemical to be eliminated from the body, assuming no additional exposure.) The half-life of PFOA has been calculated to be 5.4 years, although some studies suggest it may be shorter. (Olsen 2007)
PFOA levels in the U.S. population decreased by more than 40 percent between 2000 and 2010 and likely have continued to drop as the 2015 phase-out date drew nearer. (NHANES 2014) (Figure 2) Sampling by the Norwegian Institute of Public Health shows a very similar trend in Norwegians. (Haug 2009) This study also measured the rapid increase of PFC levels in blood serum that occurred between the mid 1970’s and 2000. (Haug 2009)
Levels of PFOS are higher, but over the last decade the average level in Americans’ blood has dropped from more than 30 parts per billion to fewer than 10 ppb, indicating that the 2002 phase-out is lowering the overall body burden. (Figure 3) (NHANES 2014)
The C8 Science Panel
The C8 Science Panel – three epidemiologists from Emory and Brown universities and the London School of Hygiene and Tropical Medicine – was tasked with researching the health effects of PFOA based on blood samples and other health data taken from almost 70,000 residents of the mid-Ohio Valley, as well as previously published studies. The mean (average) C8 level in the mid-Ohio Valley samples was 83 parts per billion, compared to 4 parts per billion in the overall U.S. population. (C8 Science Panel 2015) The goal was to “reach a judgment about whether the disease was ‘more probably than not’ linked to PFOA.” (Steenland 2014)
Parties to the settlement anticipated that the panel would do its work in a year or two at a cost of $5 million, but it took seven years and cost about $35 million. DuPont bore the cost, far more than would have been possible with funding from federal research agencies. (Steenland 2014) Panel members later wrote:
Neither the judge nor the plaintiffs were happy with the slow pace of epidemiology. The judge called us to court in 2011 to vent his frustration with our pace. He went so far as to suggest that the settling parties fire us, but fortunately they did not agree. We argued to the court, lawyers and the public that it was better to take more time and get it right. (Steenland 2014)
It’s a good thing they did. To supplement the blood sample data, the panel reviewed the medical histories of more than 16,000 valley residents, conducted a neurobehavioral study of 300 children, another of DuPont workers and others – 12 in all. It produced more than 35 peer-reviewed publications and studied probable links between C8 and 55 diseases, including 21 types of cancer, greatly expanding knowledge of the health effects of C8.
The panel’s work was groundbreaking. In the typical class-action pollution case, both sides call expert witnesses to testify “without resolving the disputed question of whether the exposure actually caused adverse health effects.” (Steenland 2014) Rather than debating the findings of studies of the general population, the panel developed detailed information on a group of almost 70,000 people with proven high exposure to the chemical, all of whom lived in the same area, had similar lifestyles and similar exposures to other pollutants. The panel’s findings are summarized in Table 2.
Table 2. Findings of the C8 Science Panel
|
Date |
Probable link |
Not a probable link |
|
Dec. 5, 2011 |
Pregnancy-induced hypertension & preeclampsia |
Birth defects Premature birth or low birth weight Miscarriage and stillbirths |
|
April 16, 2012 |
Testicular cancer Kidney cancer |
Adult-onset diabetes Other types of cancer |
|
July 30, 2012 |
Thyroid disease Ulcerative colitis |
Stroke Asthma or chronic obstructive airways Neurodevelopmental disorders in children Influenza Autoimmune diseases |
|
Oct. 29, 2012 |
High cholesterol |
Parkinson’s disease Osteoarthritis Liver disease Chronic kidney disease High blood pressure Coronary heart disease |
Source: Environmental Working Group, from C8 Science Panel Probable Link Reports
www.c8sciencepanel.org/prob_link.html
Further research
Other researchers – often basing their studies on the blood samples collected for the Science Panel – have found many other links between health harms and exposure to PFOA, PFOS and other “long-chain” PFCs (those with eight or more carbon atoms). Some of the most significant findings, all published in peer-reviewed journals, are summarized in Table 3.
Table 3. Additional Studies on Health Hazards
of C8/PFOA, PFOS and Other Long-chain1 PFCs
|
Study |
Finding |
|
Gump 2011 |
Children exposed to increased levels of PFCs may have increased impulsivity. |
|
Knox 2011 |
PFCs are associated with endocrine disruption in women. |
|
Shankar 2012 |
Exposure to high levels of C8/PFOA may increase the risk of heart disease and stroke. |
|
Grandjean 2012 |
Prenatal exposure to long-chain PFCs may reduce the effectiveness of children’s vaccines. |
|
Maisonet 2012 |
A pregnant woman’s exposure to PFOA, PFOS and other long-chain PFCs may result in baby’s low birth weight and slowed growth as a toddler. |
|
Halldorsoson 2012 |
Babies whose mothers had had higher blood levels of PFOA during pregnancy are more likely to be obese at age 20. |
|
Kristensen 2013 |
Prenatal exposure to PFOA may delay the onset of puberty. |
|
Taylor 2014 |
Women with higher levels of long-chain PFCs in blood may be more likely to experience early menopause. |
1 Perfluorinated compounds with eight or more carbon atoms
Source: Environmental Working Group, from studies as cited
Although the EPA has recommended that PFOA should not exceed 0.2 parts per billion in drinking water, the agency has not set official and enforceable standards for PFCs in water supplies. Periodically the agency requires all water utilities that serve more than 10,000 people to sample for unregulated contaminants. In the third Unregulated Contaminant Monitoring Rule, EPA included PFOA, PFOS and a number of other PFCs for testing. Through early 2015, utilities had tested nearly 23,000 samples nationwide for six PFCs. They were found at or above the minimum detectable level in 29 states, as shown in Table 4. (EPA 2015C)
Table 4. Six types of PFCs were detected in nationwide water sampling.
|
State |
Number of detections |
Highest level detected (parts per billion) |
|
Alabama |
47 |
0.18 |
|
Arizona |
29 |
0.42 |
|
California |
86 |
0.12 |
|
Colorado |
206 |
1.30 |
|
Delaware |
33 |
1.80 |
|
Florida |
27 |
0.27 |
|
Georgia |
1 |
0.01 |
|
Illinois |
3 |
0.04 |
|
Indiana |
2 |
0.08 |
|
Kentucky |
4 |
0.06 |
|
Massachusetts |
42 |
0.43 |
|
Maryland |
1 |
0.02 |
|
Maine |
4 |
0.29 |
|
Michigan |
3 |
0.06 |
|
Minnesota |
5 |
0.04 |
|
North Carolina |
32 |
0.08 |
|
New Hampshire |
4 |
0.12 |
|
New Jersey |
50 |
0.07 |
|
New Mexico |
1 |
0.01 |
|
New York |
22 |
0.17 |
|
Ohio |
7 |
0.21 |
|
Pennsylvania |
68 |
1.09 |
|
South Dakota |
2 |
0.05 |
|
Tennessee |
1 |
0.02 |
|
Texas |
7 |
0.05 |
|
Virginia |
2 |
0.02 |
|
Washington |
14 |
0.60 |
|
Wisconsin |
5 |
0.12 |
|
West Virginia |
8 |
0.09 |
|
29 states |
716 |
1.80 |
Source: Environmental Working Group, from EPA monitoring for unregulated water contaminants,
The highest number of samples with PFCs came from Colorado, all from near Fort Carson, an Army base south of Colorado Springs. The source of the contamination is unknown. The highest level in any one water supply was 1.8 parts per billion of PFOS in New Castle, Del., near DuPont’s headquarters in Wilmington. That finding prompted a shutdown of the affected wells. (Montgomery 2014) High levels of PFCs were also found near DuPont’s Chambers Works plant in Deepwater, N.J., where DuPont chemist Roy Plunkett had discovered C8/PFOA back in 1938. In 2011 DuPont settled a class-action suit over PFOA contamination of the Deepwater area’s drinking water for $8.3 million. (Dunn 2011)
The testing found that only a small percentage of America’s water supply is contaminated with PFCs. But critics – including the New Jersey Department of Environmental Protection, the American Water Works Association and even the FluoroCouncil, a global association of companies that make fluorinated chemicals – say that’s because the tests were not designed to detect lower levels of the chemical. (EPA 2011) New Jersey officials have also questioned why the testing did not include some of the next-generation PFCs its own tests had detected. (EPA 2011)
The testing methods used by New Jersey were approximately 10 times more sensitive than those specified by EPA. The less sensitive EPA tests and reporting threshold would have missed almost three-fourths of the PFC water contamination the state found in New Jersey – 80 percent if the additional chemicals New Jersey tested for were included.
The low levels of PFCs detected nationwide could mean that EPA will decline to set an enforceable standard for PFOA in drinking water, because the Safe Drinking Water Act requires that when the agency decides whether to set a standard, it must consider “the frequency and level of contaminant occurrence in public drinking water systems.” (EPA 2015D.)
The Problem With Phase-outs
In 2006, more than three years after EPA first announced its “emergency” review of PFOA, DuPont and other makers or users of PFOA in the U.S. – Arkema, Asahi, BASF, Clariant, Daikin, Dyneon (a division of 3M) and Solvay Solexis – agreed to participate in the PFOA Stewardship Program. Its stated goals reflect the slow, incremental and unenforceable process that typically follows announcement of a “voluntary” chemical phase-out. (EPA 2015B):
- To achieve, by no later than 2010, a 95 percent reduction – measured from a year 2000 baseline – in emissions of PFOA, of precursor chemicals that can break down to PFO, and of related “higher homologue” chemicals, as well as in the levels of these chemicals in manufactured products.
- To work toward eliminating these chemicals from emissions and products entirely by 2015.
EPA launched the program in 2006, but the Food and Drug Administration, which regulates chemicals in food and food packaging, did not request additional safety data from DuPont until 2008 and from BASF until 2010. DuPont, BASF and Clariant all disagreed with FDA that more testing was needed, but they agreed to stop distribution of PFOA at the end of 2011. (FDA 2012A, FDA 2012B, FDA 2012C)
The painfully slow process of reducing the public’s exposures to PFCs reflects one of the biggest flaws of the 1976 Toxic Substances Control Act – that the EPA assesses health hazards chemical by chemical, rather than as a family. Because the act places the burden on EPA to prove that a chemical is a hazard – rather than requiring the manufacturer to prove that it’s safe – the law effectively prevents the EPA from banning chemicals. Instead, the agency relies on what are called Significant New Use rules. In the absence of clear authority to ban chemicals, these rules give the agency a way to limit new uses of chemicals that may present a significant risk. EPA does not have the regulatory authority to halt ongoing uses other than negotiating a phase-out, largely on the manufacturer’s terms.
The Significant New Use rules require a company to notify EPA when it decides to manufacture for new uses or in new ways a chemical known to present a significant risk. The agency may order the company not to market the chemical or to conduct additional safety testing. Although EPA has stepped up its use of Significant New Use rules in recent years, they are still issued infrequently. It takes an average of three-to-five years for the agency to marshal the information necessary to order more testing and an additional two or more years for a company to comply. During that time the chemical may be produced and marketed without restriction. (GAO 2013) By not requiring that companies prove the safety of all chemicals before they go on the market – the precautionary principle underlying chemical regulation in the European Union, Australia and Japan – this system fails to protect Americans’ health.
After 3M disclosed to EPA what it knew about the hazards of PFOS in 2000 and agreed to a phase-out, the agency promulgated Significant New Use Rules for 88 PFOS-related compounds. (EPA 2002A, EPA 2002B) In 2007, it adopted an additional rule covering another 183 PFOS-related chemicals. (EPA 2007) However, EPA exempted so-called low-volume, low-exposure uses for which there is supposedly no viable alternative.
In January 2015, the final year of the PFOA Stewardship Program, EPA proposed an additional Significant New Use Rule for C8/PFOA and 25 other long-chain PFCs. (EPA 2015a) The rule also covers other long-chain fluorinated chemicals that could be synthesized and commercially viable.
By the end of 2015 EPA will have removed or restricted approximately 300 distinct PFCs from the market. But the process is masked in secrecy. The names of some of the chemicals subject to the 2007 rule on PFOS chemicals are kept secret as confidential business information, even though they are scheduled for phase-out. (EPA 2015B) Under the 2015 rule, the production volume and imported quantities are claimed as trade secrets for almost all the covered chemicals. This is hardly a surprise. According to a report by the Government Accountability Office, 95 percent of the information EPA receives on new chemicals contains confidentiality claims. (GAO 2013) The secrecy about what chemicals are being produced and in what volumes greatly hinders efforts by independent scientists to monitor their levels in the environment and in people and to study the possible health hazards. (EPA 2014, EPA 2015)
Enforcement is Weak, Ineffective
Section 8(e) of the Toxic Substances Control Act requires U.S. chemical manufacturers, importers, processors and distributors to notify EPA within 30 days of learning that a chemical may pose a “substantial risk of injury to health or the environment.” (EPA 2015) But the DuPont case shows that even the most serious violations do not trigger penalties sufficient to ensure compliance.
DuPont’s failure to submit studies on PFOA from 1981 to 2005 led to a $16.5 million fine, “the largest civil administrative penalty EPA has ever obtained under any federal environmental statute.” (EPA 2005) But EPA could have imposed a fine of up to $313 million – still $20 million less than the company’s third-quarter profit the previous year. (EWG 2004) The settlement gave DuPont a decade to continue using PFOA, a chemical that generated revenue of $1 billion a year. (Haber 2005) The Justice Department considered filing criminal charges against DuPont but decided in 2007 not to pursue the case. (Greenspan 2007)
Although company submissions under Section 8(e) must be made public, critical details are often kept secret as “confidential business information.” EWG reviewed more than 100 Section 8(e) submissions for fluorinated chemicals from January 2007 to March 2015. More than 85 percent did not disclose the name of the chemical involved and more than 55 percent did not disclose the name of the company. This secrecy makes it impossible for the public, state regulators or independent scientists to assess the chemical’s use and how people are exposed.
In light of the health effects reported in the studies, this lack of transparency is extremely disturbing. In studies in which the chemical’s name was withheld, reported health effects of exposure included death; maternal and developmental toxicity; degeneration and necrosis of the kidneys; chromosome aberrations; changes to the weight of the heart, kidney, liver, thymus, spleen, prostate, ovaries and adrenal glands; lethargy; and irregular breathing. This is a sampling of the submissions in which vital information was not disclosed:
- One submission detailed how rats exposed to an unknown fluorinated chemical experienced obstructed labor, decreased fertility, skeletal abnormalities in offspring and lower maternal and offspring viability during lactation. The August 15, 2007, submission did not identify the manufacturer. (Read the studies: PDF 1 PDF 2 .)
- In another reproductive study of a chemical identified only as a fluorinated aliphatic alcohol, exposed rats died, had lower body weight, experienced nursing difficulties and discolored teeth, reduced uterine weight, height, increased pup morality during lactation from dehydration, failure to nurse and nest and being cold to the touch. The study was submitted by an unidentified manufacturer on July 18, 2008. (Read the study: PDF.)
- In May 2011, 3M submitted a study of a fluorinated derivative that in test animals caused labored breathing, sluggish movement, dark eye color, delayed responses, jerky movement, extreme lung sounds and death. (Read the study: PDF.)
How Safe are the Alternatives to “Long-chain” PFCs?
With the phase-out of C8/PFOA and PFOS and hundreds of other long-chain PFCs, chemical companies and their customers have switched to alternatives with shorter carbon chains. This difference in molecular structure makes these next-generation PFCs less likely to build up in the bodies of people and animals. But like the chemicals they are replacing, they are persistent in the environment. Studies suggest that short-chain PFCS are more likely to end up in tap water because it is easier to remove long-chain chemicals by water treatment. (Eschauzier 2011)
EPA records contain disturbing indications that some of the new PFCs are as hazardous as their predecessors. But once again, the nation’s chemical regulation system lets manufacturers hide the truth. EPA does not require safety testing before new chemicals are used in products. If a company conducts studies, it doesn’t have to make them public. Manufacturers of new chemicals sometimes do publish their studies in peer-reviewed journals, but they are not required to.
A few industry papers on a select number of replacement chemicals suggest that they have lower toxicity than the C8 chemicals. (Gordon 2011, Hagenaars 2011) Similarly, studies on zebrafish indicate that developmental toxicity drops as the chain length shrinks. (Ulhaq 2013) The data EPA has reviewed suggests a similar toxicity profile for shorter-chain chemicals, because shorter-chain chemicals are cleared from the body faster. (EPA 2009)
A study of the exposure of placental cells to a four-carbon chemical, PFBS[1], associated it with disruption of the endocrine system. (Gorrochategui 2014) A study of the six-carbon chemical, PFHxS[2], found the chemical may cause nerve damage during development and affect cognitive function similar to neonatal exposure to PCBs[3], BPA[4] and PBDEs[5]. (Viber 2013)
Scientists Sound the Alarm
In November 2014, a group of prominent international scientists published the Helsingør Statement, a discussion paper raising concerns about the transition from long-chain PFCs to alternatives with fewer carbon atoms. (Scheringer et al 2014) Numerous other researchers had previously raised concerns about the replacement chemicals and the lack of information on their environmental behavior, degradation and health effects. (Chu 2014, Naidenko 2008, Zhou 2014)
The failure of the Toxic Substances Control Act to require pre-market safety testing means that in many cases hazardous chemicals have been phased out only to be replaced by others that are later found to be harmful. For example, PBDEs, or brominated flame-retardants, are being replaced by alternatives that are also toxic, persistent and build up in living things. (Butt 2014, Green Science Policy Institute 2015) Of the new perfluorinated chemicals, the Helsingør Statement (which refers to PFCs as PFASs) has this to say:
We as scientists working on the characterization of the uses, properties, analysis, environmental distribution and adverse effects of poly- and perfluorinated alkyl substances, PFASs, are concerned that long-chain PFASs are being replaced by a wide range of fluorinated alternatives for which we have only little information on production volumes, uses, properties and biological effects. Nevertheless, we do know that these replacements will be similarly resistant to ultimate degradation, i.e. persistent, in the environment as long-chain PFASs.
The Helsingør Statement was followed by the Madrid Statement, signed by more than 200 scientists from around the world, urging action to reduce the use, hazard and future consequences of PFCs. (Blum 2014) The Madrid Statement has just been published in the May 2015 issue of Environmental Health Perspectives, a prestigious peer-reviewed journal published by the National Institute of Environmental Health Sciences and the National Institutes of Health. It recommends:
- Governments should allow only essential uses of PFCs, require manufacturers to prove the chemicals are safe and provide methods to track contamination.
- Chemical companies should publicly disclose production data and studies on health effects, develop safer alternatives and label products made with PFCs.
- Manufacturers should avoid using PFCs, label products that contain PFCs and invest in the development of non-fluorinated alternatives.
- Scientists should continue to study both the old and new chemicals and work with governments and industry to compile a global inventory of all PFCs, their precursors and breakdown products, properties and health effects.
- Retailers and consumers should question the use and need for PFCs and avoid selling or buying them.
[1] Perfluorobutanesulfonic acid.
[2] Perfluorohexane sulfonic acid is categorized as a long-chain perfluoroalkyl sulfonate by EPA (EPA 2009).
[3] Polychlorinated biphenyls.
[4] Bisphenol A.
[5] Polybrominated diphenyl ethers.
Where Consumers Encounter PFCs today
Clothing
PFC coatings are added to clothing, tablecloths, car seats, upholstery, jackets, shoes, tents and more for both their water repellency as well as their resistance to oil/dirt stains. (Berger 2006, OECD-UNEP 2013, Herzke et al. 2012) These textile coatings may be both an important source of both human exposure as well as a environmental contamination. (Bringewatt 2013) Textiles account for half of U.S. consumption of polyflurorinated chemicals and of similar compounds with slightly different structures known as fluorotelomers. (EPA 2015) The textile industry originally used PFOA- and PFOS-related chemicals but has shifted to next-generation PFCs.
In 2013, Greenpeace International tested 15 samples of waterproof clothing, shoes and swimsuits and found PFCs in all but one (Brigden 2013), with six-carbon and four-carbon PFCs the most common. (Brigden 2013) Testing by a consortium of industrial, retail, academic, government and NGO partners in Europe found that water repellent coatings on apparel predominantly rely on PFCs, with C8-based chemistry still a significant portion. (SUPFES 2015) In a study of PFC levels in outdoor clothing, Greenpeace found PFCs in all products tested from the following manufacturers (listed with the brands of treated fabric they use):
- Adidas (Gore-Tex, Formation)
- Columbia (Omni-Heat Thermal Reflective, Omni-Tech Waterproof Breathable)
- Jack Wolfskin (Texapore, Nanuk 300)
- Mammut (Exotherm Pro STR)
- Patagonia (Gore-Tex)
- The North Face (Gore-Tex, Primaloft One)
Greenpeace’s campaign pressured clothing companies in Europe to form the Zero Discharge of Hazardous Compounds group. Numerous companies have gone PFC-free or have pledged to do so, including H&M, Levi’s and Puma, while Adidas has pledged to eliminate PFCs from most of its products by 2017. (Adidas 2014, H&M 2015, Levi’s 2014, Puma 2014) Noticeably absent from the Zero Discharge group are outdoor gear and sportswear companies, most of which still used PFCs, including PFOA, as of a few years ago. Patagonia, whose brand is built on environmental responsibility, says of the challenge of finding safer alternatives:
The majority of our current products that are treated with DWR (durable water repellent) now use C6 fluorocarbon-based water repellents. These are PFOS-free, but PFOA is still detectable on the treated fabric at around 100 ppb (parts per billion)… Instead of removing individual fluorinated chemicals as potential health and safety concerns are identified, it may be preferable to search for a fluorocarbon-free water repellent as a long term solution. (Patagonia 2013)
Food Wrapping
In November 2005, a former DuPont engineer named Glenn Evers, who for 22 years had worked to find new uses for PFCs in food packaging, revealed that Teflon wasn’t the only DuPont product whose hazards had been kept secret. Since at least 1981, Evers said, the company had known that PFOA chemicals similar to those in its Zonyl RP paper coating for greaseproof food wrappers and pizza boxes were bioacccumulative. DuPont appears not to have told the Food and Drug Administration, which regulates chemicals in food and food packaging and relies on information submitted to EPA under the Toxic Substances Control Act.
DuPont had known since 1987 that Zonyl RP wrappers could contaminate food with PFCs at more than three times the federal safety standard, and that two alternative chemicals leached into food at only half the federal standard. The company ignored Evers’ concerns that continuing to sell Zonyl RP was unethical. It shelved the safer alternatives and never told the FDA, its customers or the public. Evers dramatically demonstrated for news cameras how eating a hot French fry from a Zonyl RP fast food wrapper meant that consumers were putting PFCs directly into their bodies. (EWG 2005)
In the wake of the EPA fine and DuPont’s planned phase-out of PFOA, announced not long after Evers’ bombshell, fast food chains were pressured to drop PFC-coated food wrappers. Burger King stopped using fluorochemical-coated paper, and McDonald’s moved to PFOA-free coatings. (Munoz 2006) In 2008, California legislators passed a bill, sponsored by EWG, that would have banned PFOA from fast food wrappers, pizza boxes, beverage containers and other food packaging, but then-Gov. Arnold Schwarzenegger vetoed it. (EWG 2008)
As with clothing, the replacement chemicals for food wrappers and food contact materials have not been adequately tested, and detailed information about them is hidden from the public as trade secrets. Currently, DuPont advertises that it makes four shorter-chain fluorochemical coatings in New Jersey and France that are approved for use in both “oven-heated and microwaveable packaging, such as popcorn bags and fast food wrappers.” (DuPont 2015B)
FDA guidance outlines basic toxicity tests that chemical manufacturers should undertake before seeking FDA approval. (FDA 2002) Very little to no testing would be expected based on exposure estimates that FDA has completed for 46 fluorinated chemicals published in their Cumulative Estimated Daily Intake database. More than half of the 46 fluorinated chemicals in its database, including PFOA, have exposure estimates below 0.5 parts per billion in food, which would lead to an FDA recommendation that “no safety studies are recommended.” (FDA 2002) To verify the safety of the remainder of the fluorinated chemicals with higher exposure estimates, FDA would still only recommend basic genotoxicity testing to determine whether the substance causes DNA damage in bacteria or cells. (FDA 2002, FDA 2015)
While the FDA does not make public its exposure estimates, toxicity data or other testing data it evaluated in approving the use of recent PFC chemicals, the agency is required to provide non-confidential information when requested through a Freedom of Information Act request. However, confidentiality claims hide nearly all useful information, including the chemical’s identity as well as data on rates of chemical leaching into food.
EWG and the Green Science Policy Institute, a California non-profit headed by one of the authors of the Madrid Statement, identified 93 PFCs that FDA has approved for use in non-stick and grease-resistant coatings. The vast majority of these materials are intended for use on paper and paperboard containers. Fifty of FDA’s food contact approvals have come since 2000. Many of the more recent approvals are replacements for PFOS- and PFOA-based coatings. (FDA 2015) These new coatings have been submitted to FDA by a number of companies, including DuPont, Solvay, Asahi, Dailin, Greene and 3M.
In 2008 EWG reviewed the FDA safety assessments and approvals made between 2002 and 2009 for food contact substances that replaced PFOA and C-8 based PFCs. We concluded that in approving four C6-based alternatives, the agency failed to:
- adequately assess how these coatings break down
- require safety studies of the underlying C6 chemical
- back up its assessment that the replacement would not be PFOS or PFOA
- adequately consider the long-term health consequences from exposure.
Since our 2008 analysis, FDA has approved 20 additional PFC chemicals and added them to the Inventory of Effective Food Contact Substances, a database of materials approved to come into contact with food. (Wang 2013, FDA 2015) Public information on the safety of these substances is largely non-existent.
Cosmetics
Cosmetics also contain PFCs. EWG’s Skin Deep database identifies 251 products that contain PTFE, the Teflon chemical, in a wide range of products, from eye shadow to shaving cream to lip balm. Eye shadow, foundation, facial powder, bronzer and blush account for nearly 80 percent of the products with PFCs. Skin Deep® also identifies 15 other fluorinated chemicals in cosmetics.
Shamrock, a PTFE manufacturer, advertises PFC cosmetics ingredients for use in products to enhance skin feel and provide even application, water resistance and gloss. (Shamrock 2015) The Food and Drug Administration does not review the safety of cosmetics ingredients and no public studies of exposure to PFCs through cosmetic products have been completed.
Cookware
Teflon is synonymous with non-stick pans. The first Teflon-coated pan was produced in 1961. (Robbins 1986) Many non-stick pans are produced in China, where in 2004 annual production reached 100 million pans, most for export. (JingJing 2004)
The non-stick coating in Teflon pans starts to break down when the pan is heated to near 500 degrees and significantly decomposes when the temperature goes over 660. DuPont recommends that consumers make that pet birds are not kept in or near the kitchen: “Cooking fumes, smoke and odors that have little or no effect on people can seriously sicken and even kill birds, often quite quickly.” (DuPont 2015C)
In 2003, testing commissioned by EWG showed that within minutes of turning on the stove, the temperature of an empty non-stick pan would exceed 500 degrees. After five minutes the tested pans reached temperatures that would induce significant material loss and generate a range of noxious breakdown products, including four particular toxic chemicals: perfluoroisobutane, hydrofluoric acid, carbonyl fluoride and monofluoroacetic acid. (EWG 2003)
Non-stick pans with the Teflon label are currently produced using short-chain PFOA replacements. DuPont advertises GenX, a processing aid that uses an oxygen atom to reduce the length of fluorinated carbon, to generate fluorinated plastics for use in non-stick coatings for cookware. (DuPont 2010) It is possible that imported non-stick pans are covered with a non-stick coating produced using PFOA.
Holding DuPont to its Promises
Just as the phase-out of C8/PFOA did not end the global health threat from PFCs, DuPont’s $16.5 million fine and $300 million settlement did not deliver justice to the people of the mid-Ohio Valley. In many ways, it was only the beginning, and 10 years later, their fight continues.
Cleanup
In the 2005 settlement, DuPont promised to pay for cleanup or replacement of public water supplies contaminated with more than 0.05 parts per billion of PFOA. But contamination of water in the Parkersburg system, the largest in the region, was originally found to be below that threshold by a tiny amount, and DuPont moved to escape its commitment. After later tests found contamination above the threshold, Parkersburg residents filed suit to have the terms of the settlement extended to them. (Jeffersonian 2006) DuPont fought the Parkersburg residents’ claims and was able to prevail in federal court in West Virginia. The residents appealed, but in 2011 the U.S. Court of Appeals for the Fourth Circuit denied their claims. (U.S. Court of Appeals 2011)
The Little Hocking Water Association, serving a community directly across the Ohio River from DuPont’s Washington Works plant, was among the first utilities in the region to learn that its wells were contaminated with C8. In an action separate from the class-action suit, the Little Hocking utility sought to have DuPont not only clean up the water it delivers, but also the source water and the pathways the chemical traveled from the plant to the source. The utility also wanted the cleanup to cover not just C8 but other PFCs, including the next-generation alternatives, and continue in perpetuity to guard against ongoing PFC contamination of the soil and river sediment.
DuPont fought back, contending that C8 did not enter Little Hocking’s wells through the river but through smokestack emissions that seeped into the wells after rain or flooding. In March 2015, an Ohio federal court judge ruled that DuPont was liable for the utility’s claims under the Resource Conservation and Recovery Act. Damages have not been determined. (U.S. District Court 2015a)
Medical monitoring
The C8 Health Project, which gathered blood samples and information for the C8 Science Panel, gathered samples and medical histories from about 70,000 residents, far more than expected. Much credit goes to the outreach program of Brookmar Inc. of Vienna, W. Va., run by former executives of two Parkersburg hospitals. Brookmar conducted focus groups and town hall meetings, publicized the program widely in a region with no large media outlets, set up mobile testing stations and paid participants $400 apiece for their time. The program was not just a scientific success but also served to educate and unite the community. (Lyons 2007)
After the C8 Science Panel made its determinations, a medical monitoring program was set up to screen people for the six diseases linked to C8. Brookmar was to work alongside the program’s DuPont-appointed director, Michael Rozen, a partner in the New York law firm of Feinberg Rozen LLC. Rozen is known as “the special master of disaster” for his work to minimize liability costs of corporate polluters, including companies that exposed workers to asbestos and oil giant BP after the Deepwater Horizon oil spill. (Center for Justice & Democracy 2014)
According to Keep Your Promises, a mid-Ohio Valley citizens’ group, DuPont and Rozen at first agreed to work with Brookmar, but in 2013 they went to court repeatedly to argue that Brookmar’s involvement was not necessary. Brookmar grew frustrated and announced that as long as Rozen remained as director of the program, it would not participate. (Keep Your Promises 2015a)
In January 2015, Keep Your Promises released invoices showing that DuPont had paid Feinberg Rozen about $9 million for running the medical monitoring program. In contrast, the law firm had paid out just over $50,000 in medical claims to area residents. (Saulton 2015) Two days later, Michael Rozen tried to make members of Keep Your Promises leave a town hall meeting and ejected a reporter from a local TV station. (Keep Your Promises 2015b)
“What is Mr. Rozen trying to hide?” asked Harold Bock, an advisory committee member of Keep Your Promises, who resisted attempts to make him leave. “You can’t invite the whole community to an open forum and then kick out the journalists who come to cover it and the community members who come to ask important questions.” (Keep Your Promises 2015b)
The Chemours spinoff
In July 2015 DuPont will spin off its $7 billion Specialty Chemicals unit, which made C8/PFOA and now makes the replacement chemicals for Teflon and other products, to a new corporation called Chemours. U.S. Securities and Exchange Commission filings indicate that the spinoff will transfer legal liability for damage from C8 to Chemours. This could shield DuPont from full liability and allow the smaller company to claim that its assets aren’t enough to pay for the damage done in the mid-Ohio Valley and other places C8 was made or used. (U.S. District Court 2015b)
Chemours acknowledges that the Specialty Chemicals unit’s sales are declining. (SEC 2014) In September 2014, Chemours listed $298 million in environmental liabilities it will inherit, but DuPont’s own management estimates that the new company’s liability may be as much as $1 billion. (SRR 2015) Chemours’ finances, and the thousands of outstanding personal lawsuits over C8, raise the possibility that the company might go bankrupt.
It’s happened before. In one 2006 case, the oil company Kerr-McGee spun off its chemical business subsidiary, Tronox, just before merging with Anadarko Petroleum. Less than three years later Tronox filed for bankruptcy. The Justice Department brought fraud charges, charging that the spinoff did not provide Tronox with enough capital to cover its inherited liabilities and was engineered to shield the merged companies. In 2014 Anadarko settled for $5.15 billion, the largest environmental enforcement recovery payment the Justice Department ever obtained. (Checkler 2014)
DuPont on trial
Trial is scheduled for September 2015 in U.S. District Court in Columbus, Ohio, consolidating personal injury claims against DuPont by more than 2,500 residents of the mid-Ohio Valley. The court will first try two individual test cases. The first involves a woman who drank C8-contaminated water and has kidney cancer. The second, scheduled for November, involves a man who drank C8-tainted water and has ulcerative colitis.
Claims involving this much damage, affecting this many people, are more common in lawsuits against big pharmaceutical companies. It is also unusual for pollution cases to be tried with an agreement by the company not to dispute that chemical exposures at a certain level can cause a specific disease – the stipulation DuPont tried but failed to renege on.
The trial is important beyond its consequences for the mid-Ohio Valley contamination victims, because it will highlight the failures of the nation’s broken chemical safety law. DuPont maintained that the Toxic Substances Control Act did not require it to provide EPA with its secret studies. If companies can get away with withholding information on health hazards, EPA’s ability to take meaningful action under the law will always be severely compromised.
Recommendations
Ten years after DuPont was caught withholding information about the health hazards of C8/PFOA, the people and communities of the mid-Ohio Valley are still waiting for the company to make good on its promises.
For almost 70 years, DuPont has been an important institution in the region. The Washington Works plant employs more than 2,000 people and is the hub of a cluster of plastics companies that have dubbed the Parkersburg area the Polymer Alliance Zone. Many of the company’s workers proudly call themselves “DuPonters.” (Lyons 2007) Yet DuPont has repaid the community by polluting its water, knowingly exposing workers to harmful compounds that are passed along from mother to child – and now, trying to minimize the compensation its must pay. This is hardly the behavior of a good corporate neighbor.
EWG supports the Keep Your Promises campaign to hold DuPont accountable. The company should make good on the terms of the class-action settlement – and go beyond it by cleaning up any area water supply contaminated with C8 at or above the EPA’s health advisory level. In the upcoming trial the company should admit its responsibility for harming more than 2,500 area residents and pay them damages determined by the jury. DuPont must also ensure that its spinoff of Chemours doesn’t jeopardize the cleanup and compensation in the mid-Ohio Valley or anywhere else where it has polluted the environment or people.
In light of the harm done by C8/PFOA, PFOS and similar substances, it is unacceptable that companies are putting new chemicals on the market that may be no safer. Fifty years from now, will documents be uncovered revealing that DuPont, 3M or other companies knew all along that the new generation of PFCs are also hazardous? EWG joins the call of the scientists who wrote and endorsed the Madrid Statement for governments to act swiftly to assess the dangers of the new chemicals and curb their use before it’s too late.
DuPont’s malfeasance and the marketing of new chemicals that are masked in secrecy amount to severe indictments of America’s broken chemical policy.
Stronger rules on disclosure of health studies and stronger penalties for failure to disclose them could have put more pressure on DuPont and 3M to tell regulators what they knew about the hazards of PFOA and PFOS. Why didn’t the companies come clean earlier? Because they knew they could get away with it – and in many ways they did. Stronger rules would also ensure that the EPA and the FDA can adequately assess the safety of PFC replacements and enable FDA to make better decisions about the chemicals that come into contact with food.
As Congress considers reforms to the Toxic Substances Control Act, which has not been updated in almost 40 years, EWG recommends these changes to effectively generate meaningful safety data and protect Americans from the hazards of PFCs and all other dangerous chemicals:
- EPA should have the authority to ban, restrict and phase out harmful chemicals without relying on voluntary agreements.
- EPA should have authority to restrict or ban imported articles made with potentially hazardous PFCs or other chemicals.
- Chemical companies should be required to prove that chemicals are safe before they are allowed on the market.
- EPA should have the authority to require safety testing when the information provided by companies is inadequate.
- All hazardous chemicals should be tracked from production to use and ultimate disposal, and the information must be made public.
- Chemical companies should provide technologies to detect hazardous chemicals in the environment and people.
Appendix
Understanding PFCs
Per- and polyfluorinated chemicals (PFCs), more precisely defined as per- and polyfluoroalkyl substances (PFASs), are a family of chemicals used to make Teflon, Scotchgard, Gore-Tex as well as many other non-stick, stain-resistant or waterproof coatings. These chemicals share two characteristics: they take a long time to break down in the environment, and they can accumulate in the blood and organs of animals and people exposed to them. (EPA 2015) Further, the chemicals that have been studied in detail raise many similar health concerns, from cancer to liver or kidney toxicity and reproductive effects. (Scheringer 2014)
Life on earth is carbon-based. Carbon atoms form the fundamental building blocks of our bodies and those of other living organisms. Chemical compounds are built on carbon atoms bonded to atoms of such elements as hydrogen, oxygen, nitrogen, fluorine and sulfur. The carbon-fluorine bond is particularly strong, and bonding more fluorine to carbon increases the strength of all the bonds. (Kirsch 2006) The strength and stability of the bonds produces chemicals that are the slippery, repel water and dirt, resist breakdown, resist electrical current and persist in the environment for decades.
In 1938, Dr. Roy J. Plunkett, a chemist at DuPont’s laboratory in Deepwater, N.J., was trying to produce a refrigerator coolant when he accidentally synthesized PTFE. The compound was patented in 1941. (USPTO US2230654) PTFE is a synthetic plastic, or polymer, constructed from a long chain of carbon atoms with two fluorine atoms attached to each carbon atom. This two-to-one pairing produces a molecule of fluorinated ethylene. The goal in polymer production is to maximize the chain length or the number of ethylene repeats.
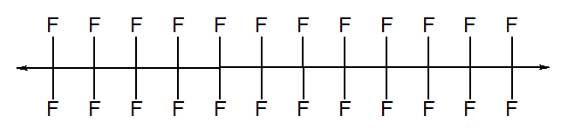
PTFE or Teflon
In 1945, DuPont trademarked PTFE as Teflon for use in industrial applications. (USPTO 1945) Subsequent trademark filings for Teflon and related chemicals chronicle its expansive usage in textiles, non-stick coatings on pans, snow shovels, bags, briefcases and umbrellas. (USPTO 1954, 1965, 1969, 2014)
PTFE has a tendency to become lumpy or bubble up. To smooth it for use as Teflon, perfluorooctanoic acid (PFOA) was added. The PFOA stabilizes the production of the polymer, and in principle should not be a component of the finished plastic. However, production and disposal result in large quantities of PFOA waste, which DuPont and other manufacturers dumped as wastewater effluent in landfills and into the air through factory smokestacks.
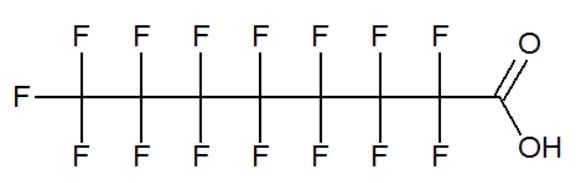
Perfluorooctanoic acid (PFOA)
Perfluorooctanesulfonic acid (PFOS) is a closely related perfluorinated chemical that also has a carbon backbone of eight atoms. Like PFOA, its discovery was an accident: In 1952, 3M chemists Patsy Sherman and Sam Smith spilled an experimental fluorochemical on tennis shoes. (Schwarcz 2004) Smith and Sherman had created the first pair of non-stick, stain-resistant shoes, and within years clothing and other textiles were widely treated with PFOS-based Scotchgard. PFOS was also used widely as a coating on food wrappers for everything from carry-out containers to microwave popcorn.
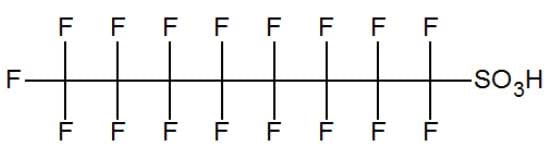
Perfluorooctanesulfonic acid (PFOS)
For replacements, the chemical companies have shifted toward shorter-chain perfluorinated compounds. The shorter-chain molecules are generally less bioaccumulative but often have decreased performance, so the U.S. market has been slow to shift to the alternatives. (Scheringer 2014) Concern has been raised that the alternatives may require higher concentrations in products to achieve the same performance, reducing the environmental or health benefits. (Scheringer 2014)
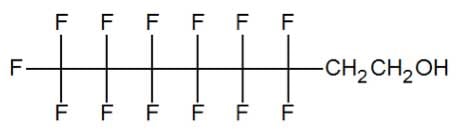
In replacing PFOA, DuPont has shifted to a shorter-chain length fluorinated chemical, known as a six-two fluorotelomer alcohol, indicating that the fluorinated carbon length is six atoms long. Keeping the fluorinated section of the molecule less than eight carbon atoms long assures that the chemical will not break down to PFOA or PFOS in the environment. (DuPont 2015A)
DuPont replacement for PFOA
3M developed an alternative to PFOA, abbreviated as ADONA, by breaking up the fluorinated carbon backbone with oxygen atoms. (Gordon 2010) This approach limits the maximum length of a breakdown product to three carbon atoms or less.
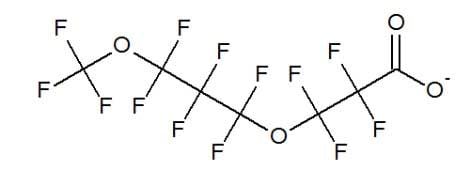
3M replacement for PFOA
Since 2000, EPA has received and reviewed pre-manufacture notices for more than 150 different chemicals to replace the longer-chain PFCs. (Krasnic 2014) Neither the chemical companies nor EPA have provided information on how the safety of these replacement chemicals has been verified or how many of these chemicals have already found their way onto the market.
The PFOS replacements include chemicals that incorporate PBSA, a four-carbon chain sulfonamide. It has been reported that the salts of the 6:2 fluorotelomer sulfonic acids are also being used as replacements. (Chu 2014, Wang 2013 B) The main reported in-vitro metabolite of ScotchGard post-2002 is PBSA. (Chu 2014)
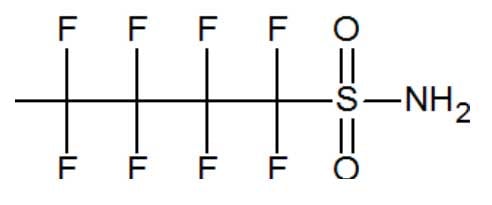
PBSA





