
View and Download the report here: In the Dust
High Levels of Toxic Fire Retardants Contaminate American Homes
In the first nationwide tests for brominated fire retardants in house dust, the Environmental Working Group (EWG) found unexpectedly high levels of these neurotoxic chemicals in every home sampled. The average level of brominated fire retardants measured in dust from nine homes was more than 4,600 parts per billion (ppb). A tenth sample, collected in a home where products with fire retardants were recently removed, contained more than 41,000 ppb of brominated fire retardants — twice as high as the maximum level previously reported by any dust study worldwide.
Like PCBs, their long-banned chemical relatives, the brominated fire retardants known as PBDEs (polybrominated diphenyl ethers) are persistent in the environment and bioaccumulative, building up in people's bodies over a lifetime. In minute doses they and other brominated fire retardants impair attention, learning, memory and behavior in laboratory animals.
EWG's test results indicate that consumer products, not industrial releases, are the most likely sources of the rapid buildup of PBDEs in people, animals and the environment, which has been documented by tests from Europe to the Arctic. Scientists now recognize that indoor environmental contamination, including contaminants accumulating in household dust, pose a substantial health risk to the population. Our findings raise concerns that children may ingest significant amounts of toxic fire retardants via dust, and indicate that the impending federal phase-out of two PBDEs doesn't go far enough to protect Americans.
Two of the three main PBDE products in use, Penta and Octa, will be taken off the U.S. market at the end of 2004. The fire retardants industry has strongly resisted the regulation of the third product, Deca, maintaining that it is not harmful despite mounting evidence that shows Deca is toxic, detected widely in the environment, and can break down to more harmful forms, including those being phased out.
In half of the homes EWG sampled, we found the predominant PBDE present was the type found solely in the Deca product. We also found important new evidence of PBDEs' chemical breakdown, underscoring the fact that current federal and state efforts to get rid of harmful PBDEs may be in vain if they don't include Deca.
In September 2003, nationwide tests by EWG found record levels of PBDEs in the breast milk of American mothers. This follow-up study of household dust includes 10 of the 20 participants from the breast milk study, and is the first study to compare the concentrations of fire retardants in people and in their homes. Although PBDE concentrations in dust are much higher than those found in food, water, air or soils, we found no correlation between PBDE levels in house dust and in breast milk. This finding highlights the important yet still unanswered question of whether some people absorb more PBDEs than others, metabolize these chemicals differently, or are slower to eliminate them.
High levels of PBDEs were found in all ten homes tested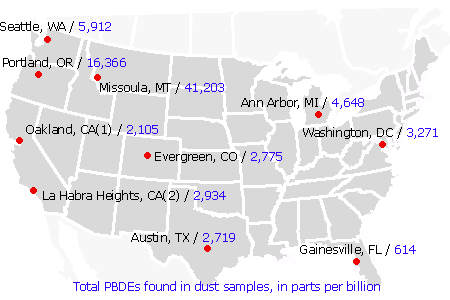
It is no surprise that American homes are contaminated with PBDEs. They are added to thousands of everyday products, including computers, cars, TVs and furniture. But our tests show the surprising degree to which these chemicals are escaping from consumer products. The PBDE concentrations we measured in house dust are much higher than levels previously reported in people, animals or the environment, and also pose a more direct risk of exposure to people, especially children, who continually ingest or inhale dust.
Penta and Octa will be banned by law in the European Union this year, in California in 2008, and under an Environmental Protection Agency agreement with PBDE makers, will not be manufactured in the U.S. beginning next year. In addition, measures to study, regulate or ban one or more PBDEs have either been enacted or are under consideration in Hawaii, Maine, Massachusetts, Michigan, New York and Washington state. Regulatory or legislative proposals to add Deca to the EU and California bans are under consideration this year. A bill introduced in the U.S. Congress last month would ban Penta and Octa in 2006, and directs EPA to identify and ban proven precursors (such as Deca) within three years.
The EPA should not wait. It is no longer possible to ignore the evidence that Deca poses a threat to health and the environment. Deca was found in 16 of 20 breast milk samples analyzed by EWG in 2003. In our latest tests, Deca was found at equal or greater concentrations in American homes than the PBDEs subject to phaseout, to which all evidence indicates it is a precursor. Deca's toxicity to laboratory animals, its presence in the human body and its breakdown to more toxic forms demand prompt action.
EWG recommends:
- The EPA must promptly phase out all PBDEs, including Deca.
- All consumer goods containing PBDEs must be labeled so that consumers may choose fire retardant-free products.
- The EPA and the Consumer Product Safety Commission must issue clear advice American consumers on the safe use and disposal of products containing PBDEs.
- EPA must rigorously test potential replacement fire retardants to ensure that they are not persistent, bioaccumulative or toxic. Redesigning products to reduce the need for chemical fire retardants is safer than substituting new compounds that may be later found harmful.
View and Download the report here: In the Dust
Brominated Fire Retardants
Brominated Fire Retardants: Persistent Global Pollutants
As highly flammable synthetic materials have replaced less-combustible natural materials, chemical fire retardants have become common in consumer products. One of the most widely used belong to a class of chemicals known as brominated fire retardants.
Fire retardants are the single largest end use of bromine worldwide. [1] About 449 million pounds of brominated fire retardants were used worldwide in 2001, and the market is growing by four percent annually. [1, 2] The brominated fire retardants known as PBDEs (polybrominated diphenyl ethers) are the most widely used in North America, which used about 149 million pounds of PBDEs in 2001, half the world total.
Global Use of Commercial PBDE Products in 2001
(in thousands of pounds)
| Commercial PBDE Product |
Americas | Europe | Asia | Other | Total | Percent used in the Americas |
|---|---|---|---|---|---|---|
| Deca | 54,010 | 16,760 | 50,710 | 2,315 | 123,700 | 44% |
| Penta | 15,650 | 331 | 331 | 221 | 16,530 | 95% |
| Octa | 3,307 | 1,345 | 3,307 | 397 | 8,356 | 40% |
Source: [1]
PBDEs are in thousands of everyday products, including electronics equipment, lighting, wiring, building materials, textiles, furniture and industrial paints. [3] These fire retardants often make up a considerable proportion of product weight: Plastic can be up to 15 percent PBDEs and polyurethane foam up to 30 percent PBDEs. [4, 5]
Two of the largest manufacturers of brominated fire retardants are Great Lakes Chemical Corp. of West Lafayette, Ind., and Albemarle Corp. of Richmond, Va. In 2002, Great Lakes reported total sales for all products of $1.4 billion, up 4 percent from the previous year. Albemarle reported sales of $980 million, up 7 percent. [6, 7] The main areas of bromine production in the world are southeastern Arkansas, where Great Lakes and Albemarle pump it from underground pools of brine, and Israel, where a company named Dead Sea Bromine extracts it from the briny inland sea. A chemical industry journal describes the global trade in brominated chemicals as "an oligopoly controlled by Albemarle, Great Lakes and the Dead Sea Bromine Group." [8]
Products Often Containing PBDEs
(Bold indicates major uses)
| Materials used in | PBDEs | Examples of consumer products | ||
|---|---|---|---|---|
| Deca | Octa | Penta | ||
| Plastics | x | x | x | Computers, televisions, hair dryers, curling irons, copy machines, fax machines, printers, coffee makers, plastic automotive parts, lighting panels, PVC wire and cables, electrical connectors, fuses, housings, boxes and switches, lamp sockets, waste-water pipes, underground junction boxes, circuit boards, smoke detectors. |
| Polyurethane foam | x | Home and office furniture (couches and chairs, carpet padding, mattresses and mattress pads) automobile, bus, plane and train seating, sound insulation panels, imitation wood, packaging materials. | ||
| Textiles | x | x | Back coatings and impregnation of home and office furniture upholstery, industrial drapes, automotive, aircraft and train seating. | |
| Paints and lacquers | x | x | Marine and industry protective lacquers and paints. | |
| Rubber | x | x | Conveyor belts, foamed pipes for insulation, rubber cables. | |
Sources: [3], [4]
PBDEs are similar in structure to PCBs, the family of highly persistent and bioaccumulative toxicants that were banned in the 1970s after the discovery of widespread pollution which persists to this day. Like PBDEs, they have been found in people, animals and the environment worldwide. And like PCBs, scientists have found that exposure to minute doses of PBDEs at critical points in development can cause deficits in motor skills, learning, memory and hearing, changes in behavior, and decreased sperm count. [9, 10, 11, 12, 13, 14]
In 1999, Swedish researchers reported that PBDE levels in women's breast milk had increased 60-fold between 1972 and 1997. [15] Similar dramatic increases were documented in California harbor seals, ringed seals from the Arctic, gull eggs from the Great Lakes and human blood from Norway. [16, 17, 18, 19] PBDE pollution has been found essentially everywhere scientists have looked: in the tissues of whales, seals, birds and bird eggs, moose, reindeer, mussels, eels, and fish; in human breast milk, hair, fat and blood; in twenty different countries and remote areas such as the North Sea, the Baltic Sea and the Arctic Ocean. [21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31] In response to the evidence, this year the European Union will ban the PBDE mixtures known as Penta and Octa (named for the typical number of bromine atoms in the mixture's chemical constituents).
Levels of fire retardants rose dramatically in
Swedish breast milk from 1972 to 1997
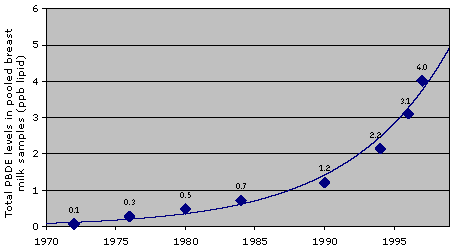
Source: [15]
PBDE levels detected in U.S. studies are consistently much higher than levels anywhere else in the world. In September 2003, EWG tests found that the median level of PBDEs in the milk of 20 first-time American mothers was 30 times that found in recent European studies. [30] Similar levels have been found in several other U.S. studies of infant cord blood, women's blood, adipose tissue and breast milk. [31, 32, 33, 34] In 2003 evidence of the rapid buildup of PBDEs prompted California to follow Europe's lead and ban Penta and Octa beginning in 2008. Within months of the enactment of the California ban, the EPA reached agreement with U.S. PBDE makers to stop manufacturing Penta and Octa by 2005.
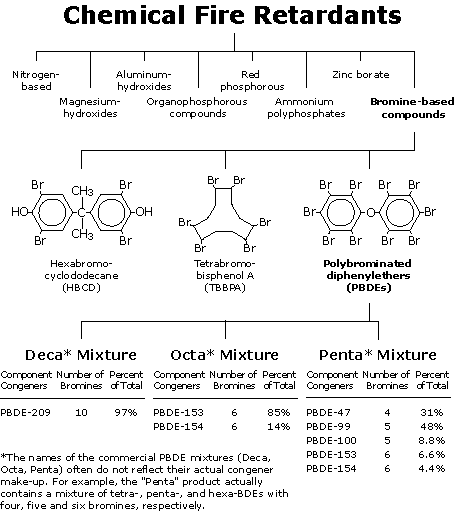
Clearly Penta and Octa are on the way out worldwide. But the chemical industry is waging a fierce fight to retain the use of the third major PBDE compound, Deca, despite mounting concerns about its human health effects and its tendency to breakdown into other more toxic and bioaccumulative PBDEs. Deca is the most widely used of the PBDEs, making up 83 percent of the global market and 74 percent of the U.S. market. [1, 2] Most of the Deca produced worldwide is manufactured by Albemarle, Great Lakes Chemical and the Dead Sea Bromine Group. [35] U.S. chemical manufacturers and users alone report more than 1.4 million pounds of Deca emissions in 2001,— more than double the amount that was released in 1988, the first year records were kept. [36]
Deca is primarily used in the plastic housings of electronic equipment such as computers, televisions, and office machines, and also in plastic auto parts, lighting panels, electrical connectors and fuses. About 10 percent of the Deca produced is used in the textile industry, which applies the chemical to the upholstery of home and office furniture, car, plane and train seating, and industrial drapes. A small amount of Deca is also used for industrial paints and lacquers. Rather than chemically binding to the treated material like some other fire retardants, Deca is simply mixed in during manufacturing, making it more likely to leach out.
In California, Europe and most other places that have tried to enact PBDE regulations, chemical manufacturers lobbied heavily to retain the use of Deca, arguing that it does not bioaccumulate because its molecules are too large to be absorbed by organisms. [37] Recent research has proven otherwise. Despite analytical barriers that make its detection difficult, Deca has now been found in wildlife worldwide, including fish, seals, porpoises, dolphins, star fish, hermit crabs, mussels, shrimp, birds and bird eggs. [38, 39] In some cases the levels detected have been quite high: Dolphins in the North Sea and peregrine falcon eggs in the U.K., for example, had levels measuring 318 and 828 ppb (lipid), respectively. [38, 39]
Deca-BDE has been detected in people and numerous wildlife species
| Species | Location |
Max deca-BDE level detected |
|---|---|---|
| Peregrine falcon egg | United Kingdom |
828 |
| Peregrine falcon egg | Sweden |
430 |
| Dolphin | North Sea |
318 |
| Human blood (occupational exposure) | Sweden |
278 |
| Human blood (general population) | United Kingdom |
241 |
| Harbor seal | North Sea |
160 |
| Mysid shrimp | Western Scheldt |
93 |
| Common tern eggs | Maasvlakte |
70 |
| Bream | Germany |
37 |
| Harbor porpoise | North Sea |
26 |
| Star fish | North Sea |
8.9 |
| Hermit crab | North Sea |
7.0 |
| Common Kestrel | United Kingdom |
5.5 |
| Grey heron | United Kingdom |
4.5 |
| Chicken egg | United States |
3.4 |
| Cod | North Sea |
2.5 |
| Barn owl | United Kingdom |
2.5 |
| Red kite egg | United Kingdom |
2.3 |
| Eurasian sparrow hawk | United Kingdom |
2.2 |
| Mussels | Japan |
1.4 |
| Montagu's harrier egg | United Kingdom |
1.3 |
| Great crested grebe | United Kingdom |
1.2 |
Source: [38, 39, 81]
It is now clear that Deca is also getting into our bodies. Scientists have detected it in human hair, fat, blood and breast milk. In our breast milk study EWG found Deca in 16 of 20 first-time U.S. mothers in concentrations as high as 1 ppb, while another study released at the same time found it in the breast milk of eight of 23 Texas women at a maximum of 8 ppb. [30, 34] Much higher concentrations have been found the blood of the general population of the U.K. and workers in Sweden (up to 241 and 278 ppb, respectively). [39, 38] These findings refute the chemical industry's claim that it is virtually impossible for Deca to enter women's bodies, and even more unlikely for her to pass it on to her child via her breast milk. [37]
New research also indicates that Deca is more toxic than previously thought, causing some of the same effects on newborn rats and mice as Penta and Octa. [40] Equally importantly, scientists have found that although Deca is highly persistent under some conditions, when exposed to sunlight it breaks down — that is, it loses bromine ions — to become the forms of PBDEs which are more toxic and more likely to accumulate and persist in people. [41, 42, 43, 44, 45] This finding is consistent with the fact that structurally similar chemicals like PCBs also break down in the environment.
High Levels of PBDEs Found
In September 2003, EWG reported record levels of PBDE contamination in the bodies of 20 American mothers. Later that month we initiated a dust study to determine the extent of PBDE contamination in the indoor environment. We asked ten women from the breast milk study to collect samples of dust from their homes. These participants were selected to be a representative sample of the original group with respect to geographical location, PBDE body burden, age and occupation. Women who had moved since submitting their breast milk samples were excluded from the follow-up study. No one reported occupational exposure to foam or plastics, except the use of computers in an office setting.
The ten study participants vacuumed their house as they normally would and sent EWG either the dust bag from their vacuum cleaners or, if they owned vacuum cleaners without bags, emptied the cartridge of their machine into a clean zip-lock bag. The women also filled out a questionnaire on the model and make of their vacuums, how many times they had used their machine since last changing the bag or emptying the canister, and the number of rooms in their house with carpet. Samples were sent to a certified laboratory for analysis.
The results were striking. Each of the ten dust samples contained high levels of PBDEs, with concentrations exceeding 3,000 ppb for half of the study homes and averaging 4,629 ppb. Overall, PBDE levels ranged from 614 to 16,366 parts per billion. We chose to treat one participant's sample separately because she had used her vacuum to clean up polyurethane foam residues when she removed carpet padding, two mattress pads, and an uncovered foam cushion from her home. Her sample contained 41,203 ppb of PBDEs — a level twice as high as reported in any previous study.
Overall, the PBDE levels we found in house dust, though very high, are actually in line with what has been reported in other studies. A study of 100 homes in the UK, for example, measured average levels of 10,543 ppb in dust, with a maximum of 20,505 ppb. The only other U.S. study to look at house dust, in Cape Cod, Mass., found an average PBDE concentration of 3,669 ppb and a maximum of 11,426 ppb [46, 47]
There are 209 chemically distinct variations, or congeners, of PBDEs; the commercial compounds sold as Penta, Octa and Deca are mixtures of several different congeners. Though we tested for thirteen different PBDE congeners, just three of these — PBDE-47, -99 and -209 — comprised 90 percent of the makeup by weight of our dust samples. The two major ingredients in Penta, PBDE-47 and -99, each accounted for 24 percent on average. But PBDE-209, the major ingredient in Deca, was the dominant congener found, making up 42 percent of the samples. Levels of Deca averaged 2,394 ppb, ranging from less than 400 ppb to 7,510 ppb.
High levels of PBDE were found in dust from 10 American homes
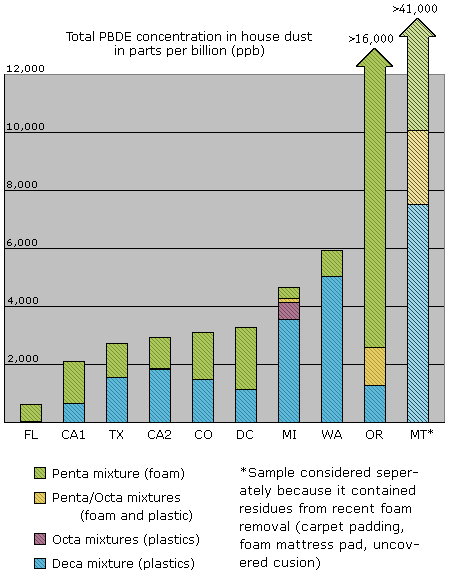
One factor may partially explain the wide variation we observed in dust concentrations. Both of the two participants with the highest concentrations of Penta in household dust reported atypical uses of polyurethane foam in their homes. One participant had reupholstered her couch cushions several years before the sample was collected; the other had used her vacuum to clean up the following polyurethane foam products: carpet padding from one room, two mattress pads and a uncovered foam cushion.
Evidence of breakdown in homes
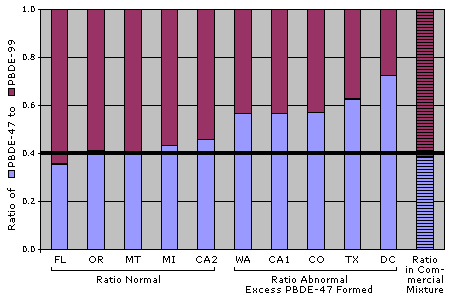
Another interesting finding of our study relates to the ratio of the two main PBDE congeners found in the Penta product: While the Penta mixture contains sixty percent PBDE-99 and forty percent PBDE-47, five out of our ten dust samples contained a significantly higher percentage of PBDE-47 -— and one home contained 50 percent more PBDE-47 than expected. In other words, half of the homes tested had an abnormal ratio of these chemicals as compared to the ratio found in the commercial Penta product.
Because PBDE-99 has five bromines while PBDE-47 only has four, our data suggests that PBDEs are breaking down inside study homes. This is of particular concern because PBDE-47 is more bioaccumulative than PBDE-99 or -209. In EWG's breast milk study, for example, we found that women had at least twice as much PBDE-47 as PBDE-99 in their bodies. This finding also underscores the concern for Deca also breaking down in people's homes and the environment. With such high levels of Deca found in house dust, even if the breakdown occurs slowly or to a small degree, Deca could nevertheless be an important source of exposure to the more toxic and bioaccumulative forms of the PBDEs.
The ratio of PBDE-47 to PBDE-99 in dust samples suggest that PBDEs are breaking down into more toxic forms in many homes.
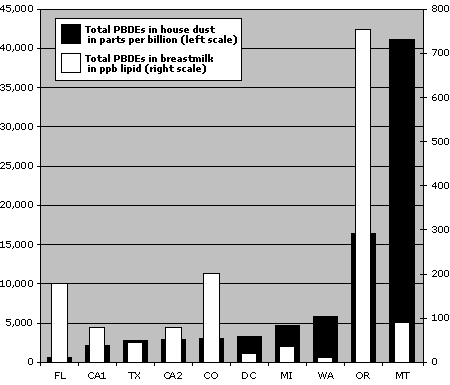
Dust and milk levels not related
Given that EWG analyzed only one dust sample per household taken at a single point in time, it was not surprising that we found no direct relationship between concentrations of PBDEs in a woman's body and in her home. (This was true for each type of PBDE, when analyzed together or alone.) PBDE concentrations in dust did not appear to be affected by the number of electronic appliances or computers, foam furniture, or recent remodeling. Diet, age, occupation and other demographic factors also failed to explain the wide variation in body burdens found by EWG in our 2003 study of PBDEs in women's breast milk.
The sources of PBDE contamination measured in human beings are currently unknown. Animal fats are the major source of PCB and dioxin exposure, and are suspected as an important source of PBDEs as well. However, several factors make it difficult to quantify PBDE exposure. First, PBDE body burdens accumulate after years of exposure. Ideally, exposure assessment would measure concentrations of fire retardants in a woman's diet, and from the air and dust in her recent homes, offices, and vehicles. Also, no study has looked at individuals' rate of uptake, metabolism and excretion of these chemicals, which is likely to play a large role in the highly variable concentrations observed in human beings. Further research will help clarify the role of household dust as direct route of exposure for children and mothers. But our preliminary comparison shows that dust ingestion may be a particularly significant route of exposure for young children.
PBDE levels in house dust vs. breast milk
Dust and Indoor Pollution
Some researchers refer to house dust as an "indoor-pollution archive" or a "long-term accumulative sample" of contaminants, because contaminants accumulate in house dust over time. [48, 49, 50] Ordinary house dust is a complex mixture of pet dander, fungal spores, particulates from indoor aerosols, soil tracked in by foot traffic, VOCs and traces of metals. [51] House dust is not as exposed to moisture and sunlight which typically facilitate the breakdown of chemicals in the environment. Therefore many contaminants degrade more slowly in dust, or not at all. [52] For example, although the toxic pesticide DDT was banned in the 1970s, it was found in detectable levels in 65 percent of samples collected in Massachusetts homes and 70 percent of German homes. [47, 53]
Numerous studies have now found that the indoor environment contains higher levels of toxic fire retardants than are found outside. [54, 55] PBDEs mix with house dust as foam furniture degrades or electronic products emit chemicals through off-gassing. The high concentrations we found suggest that degrading consumer products, not emissions from industrial facilities, are the likely source of fire retardants measured in people, fish and wildlife in far-flung regions of the world.
PBDEs in dust: High levels found in homes and offices worldwide
| Study Location(s) | Number of samples | Total PBDEs (ppb) | Deca (ppb) |
|||
|---|---|---|---|---|---|---|
| Average | Range | Average | Range | |||
| United Kingdom | 10 pooled samples of 10 houses each | 10,543 | 4,254 - 20,505 | 9,820 | 3,800 - 19,900 | |
| EWG study | 10 houses | 4,629 | 614 - 16,366 (41,203) | 2,394 | < 400 - 7,510 | |
| Cape Cod | 5 houses | 3,699 | 1,412 - 11,426 | 1,232 | 916 - 1,472 | |
| Germany | 25 houses | 1,807 | 145 - 27,008 | 1,394 | 137 - 19,100 | |
| Norway, Finland | 2 houses | 267 | 129 - 405 | 180 | 100 - 260 | |
| Netherlands, Finland, Sweden, Italy, Denmark | 7 Parliament buildings | 2,371 | 437 - 7,100 | 2,129 | 330 - 6,900 | |
| Netherlands | 3 internet providers | 405 | 311 - 546 | 360 | 260 - 490 | |
Sources: [46, 47, 107, 108]
We also found PBDE elevated concentrations relative to other contaminants in house dust. In a recent study, researchers tested dust from 120 homes in Cape Cod, Mass., for 63 common chemicals. The average PBDE levels in EWG's dust samples were higher than most pesticides, PCBs, parabens and polyaromatic hydrocarbons found in Cape Cod. [47] More than 80 percent of the contaminants found in the Cape Cod study were found in lower mean concentrations than any of the three major PBDE congeners in EWG's dust samples.
Scientists now recognize that indoor environmental contamination poses a substantial health risk to the population. Pollution levels indoors, where Americans spend 90 percent of their time, are much higher than outdoors. [56] Efforts to examine and reduce indoor pollution have focused on a handful of agents — secondhand smoke, radon, lead paint, pesticides, formaldehyde and other volatile organic compounds (VOCs). But very little is known about the risks associated with exposure to toxic chemicals in house dust. [56]
Lead was the first environmental contaminant for which dust was found to be an important pathway for exposure. Today, many epidemiological studies have shown strong relationships between the concentration of lead in house dust and the blood of those in the home, and have confirmed that lead-contaminated house dust is a major source of lead exposure in children. [57, 58, 59]
Exposure to contaminated dust is particularly of concern for children. Not only do children spend a lot of time on floors and carpets where dust accumulates, they frequently put their hands and other objects in their mouths, increasing the ingestion of dust and the contaminants in it. [50] Children also inhale more than adults relative to body weight, and typically have more skin surface area exposed, which is important since some contaminants can be absorbed directly through the skin. [60] Overall, infants and toddlers ingest about twice as much dust as adults per day. [61] The EPA recommends that scientists estimate 100 mg dust ingestion per day for children ages 1 to 4, and 400 mg for highly exposed children. [62] Some children who compulsively eat dirt (a phenomenon known as pica behavior) may ingest ten or more times this amount. [61]
EWG's calculations show that dust is likely to be a more important PBDE exposure route for children than food. Six studies have estimated PBDE exposure in a daily adult diet for European and Canadians, reporting concentrations ranging from 13 to 213 ng/day. [55, 63, 64, 65, 66, 67] Figures for the US diet are believed to be within a similar range. [68] By comparison, when EWG calculated children's daily PBDE ingestion via dust (using median and high-end concentrations measured in our study and EPA's estimates for childhood dust ingestion), our mid-range estimate was 327 ng/day; our high-end estimate was twenty times this amount. [62] And children ingest a much smaller quantity of food than adults, this comparison is actually an overestimate of children's PBDE exposure from food.
Dust is a more significant source of
PBDE exposure for children than food
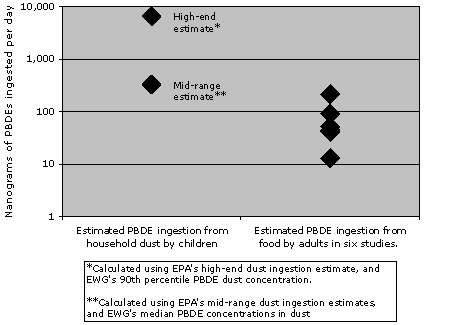
Sources: [55,62,63,64,65,66,67]
In addition to being more exposed to indoor environmental contaminants via dust, children are often more susceptible to the effects of the contaminants themselves. In children, toxins have more opportunity to cause lasting damage because even small perturbations in hormone levels at the wrong time can disrupt normal brain and organ development. Age-related differences in human metabolism can also make infants and children more sensitive to chemical exposures than adults. For example, a comparison of the half-lives (a measure of how fast a chemical leaves the body) of 45 different pharmaceuticals in young children and adults found that on average it takes newborn babies 3 to 9 times longer to eliminate half of the administered dose. [69]
House dust is a reservoir for many environmental toxins: Selected results of 3 major studies
| Chemical Name | House dust concentration (ppb or ng/g) |
Common Uses | Number and Location of Homes Sampled | Source | |
|---|---|---|---|---|---|
| Median | Max | ||||
| TBBPA, tetrabromobisphenol A | ND | 340 | Fire Retardants | 110 homes in the UK |
[46] |
| HBCD, hexabromocyclododecane | 3,200 | 6,900 | |||
| Tris breakdown product, 2,3-dibromo-1-propanol | ND | 42,800 | 88 homes in Cape Cod |
[47] | |
| bisphenol A | 0.8 | 18 | Plastics additive | 118 homes in Cape Cod | [47] |
| dibutyltin | 519 | 1,300 | 119 homes in the UK | [46] | |
| dioctyltin | 63 | 545 | |||
| monobutyltin | 1350 | 2,800 | |||
| monooctyltin | 349 | 1,300 | |||
| tributyltin | 50 | 759 | |||
| benzyl butyl phthalate | 45.4 | 1,310 | PVC plastic, personal care product additive | 119 homes in Cape Cod | [47] |
| bis(2-ethylhexyl) adipate | 6.0 | 391 | |||
| bis(2-ethylhexyl) phthalate | 340 | 7,700 | |||
| diethyl phthalate | 5.0 | 111 | |||
| di-N-butyl phthalate | 20.1 | 352 | |||
| bendiocarb | 0.4 | 318 | Pesticide | 362 homes, nine states |
[109] |
| carbaryl | 0.4 | 1,160 | |||
| chlorpyrifos | 0.6 | 324 | |||
| diedrin | 0.3 | 139 | |||
| DDT | 0.3 | 10 | 119 homes in Cape Cod | [47] | |
| methoxychlor | 0.2 | 13 | |||
| pentachlorophenol | 0.8 | 8 | |||
| piperonyl butoxide | 0.4 | 624 | |||
| trans-permethrin | 0.4 | 98 | |||
| methyl paraben | 1.0 | 8 | Preservative: food, cosmetics | 118 homes in Cape Cod | [47] |
| 4-nonylphenol | 2.6 | 9 | Industrial detergent, personal care products | 118 homes in Cape Cod | [47] |
| nonylphenol diethoxylate | 5.3 | 49 | |||
| nonylphenol monoethoxylate | 3.4 | 16 | |||
Health Risks of Deca
Most research on the toxic effects of PBDEs has centered on those congeners with five or fewer bromines — the predominant chemicals in Penta. Since these less-brominated PBDEs are readily absorbed by the body, slowly eliminated, highly bioaccumulative, and structurally similar to PCBs, it wasn't that surprising when scientists began to find that low doses of these PBDEs could disrupt the thyroid hormone balance and interfere with neurodevelopment in laboratory animals. [37, 9, 70, 71, 72, 73, 74, 75]
The initial scientific view of Deca was quite different. It was thought that Deca was too large to be absorbed, toxic only at very high doses, and not found in living things. But as more independent scientists examined Deca more closely, this dogma crumbled. Because Deca is challenging to test for, most researchers didn't bother. As one scientist explained in a recent paper: "The 'absence' of [Deca] in the ambient population is likely a result of analytical bias; that is, most studies of the ambient population did not include [Deca] as one of the analytes of interest." [76]
Since researchers have started looking for it more consistently, Deca has been detected in dozens of wildlife species from around the globe as well as human hair, fat, blood and breast milk. Although levels of Deca found in animals and humans are much lower than other PBDEs — on average, five percent of total PBDE concentrations — this is not universally true. Some studies of human tissue found Deca made up as much as one-fourth of PBDEs in people who were not exposed on the job, and as much as 40 percent in people who were occupationally exposed. [76] In Europe, Deca has been found in birds at up to 530 ppb, and in falcon eggs at up to 828 ppb. [38, 39]
According to the EU's draft risk assessment, "The potential for uptake of [Deca] by mammalian systems may be higher than indicated by many of the previous laboratory studies," and the absorption "could be quite large." [39] One study, found that 13.5 percent or more of Deca persisted in the brain, liver, and hearts of mice 24 hours after dosing. [40] Another study indicates that rats absorb 10 percent to 65 percent of a single dose of Deca. [77]
If Deca were truly non-toxic, as the fire retardant industry maintains, or only harmful at very high levels, the fact that it can be absorbed by living things might not matter. But new research is challenging this commonly held notion. A study exposing mice to a single low dose (2.2 mg/kg) of Deca during a "defined critical phase of neonatal brain development" caused "irreversible changes in adult brain function" which worsened with age. [39, 40] These mice, when tested later as adults, showed "neurobehavioral derangements." [40]
Harm at just one dose?
| PBDE congener | Part of what commercial mixture | Test animal | PBDE concen- tration in fat tissue |
PBDE dose (mg/kg-day) |
Toxic effect | Source |
|---|---|---|---|---|---|---|
| PBDE-209 | Deca | Mice | Not measured | 2.2 (single dose) | Caused aberrations in spontaneous motor behavior and habituation capability that worsened with age | [40] |
| PBDE-99 | Penta | Mice | 12 ppb in brain lipid* | 0.8 (single dose) | Effects on learning and memory, spontaneous motor behavior and habituation capability that worsened with age | [9, 10] |
| Commercial Penta Mixture DE-71 | Penta | Mice | Not measured | 0.8 (single dose) | Significant decrease in thyroid hormone (T4) levels | [70] |
| PBDE-99 | Penta | Rats | Not measured | 0.06 (single dose) | Decreased sperm count | [95] |
| PBDE-99 | Penta | Rats | Not measured | 0.06 (single dose) | Changes in subcellular structure of ovaries | [110] |
Interestingly, newborn mice exposed during a different phase of development showed no such effects, highlighting the importance of timing in determining whether exposure to PBDEs may cause adverse health effects later in life. Researchers from two different labs found similar neurobehavioral effects when they exposed newborn mice and rats to low doses of tetra and penta PBDEs during certain periods of development. [9, 10, 78] Deca is the only commercial PBDE mixture that has been tested for its ability to cause cancer. In a single study 15 years ago, researchers found high doses of Deca given to rats and mice caused liver, thyroid and pancreas tumors. [79]
The most important question about Deca, however, is its breakdown to less-brominated forms of PBDEs. While there is still debate about whether low doses of Deca are harmful, there is no question that low doses of Penta are. Studies from as far back as the early 1970s clearly show that Deca degrades into less-brominated PBDEs after exposure to sunlight. But the implications of such findings were not recognized until much more recently. [80]
The EU draft ecological assessment concludes: "Available data on photodegradation of [Deca] clearly show that the substance photodegrades under a range of conditions." [81] A leading toxics researcher, Linda Birnbaum of the Environmental Protection Agency, agrees:
"There is now good evidence that Deca can and does break down both in the environment and in fish, as well as in rats. . . . We really don't know if the breakdown products are contributing to the body burdens of these congeners in biota and people. We also don't know the toxicity of these specific congeners, although at least one of them . . . has been shown to be developmentally neurotoxic by studies in Sweden." [82]
Today, numerous studies confirm that Deca can debrominate under a wide range of conditions. [41, 42, 43, 44, 83, 84, 85, 86, 87] One study found that Deca can degrade by 50 percent in as little as 15 minutes, while another found that after five days of exposure to sunlight just six percent of the chemical remained undegraded. [41, 85] These rates are probably much higher than what occurs in the real world, but the bottom line is that these studies found that Deca degradation products were "readily formed on all matrices." [86]
Key Debromination Studies
| PBDE congeners tested | Part of which commercial mixture(s) | Study methodology | Source |
|---|---|---|---|
| Study findings | |||
| PBDE 209 | Deca | Deca was dissolved in toluene (solvent) and exposed to artificial ultra-violet (UV) light for four hours. | [87] |
| Deca degraded to nona- to penta-brominated PBDEs. Some dioxin-like compounds (polybrominated dibenzo-furans or PBDFs) were also formed. | |||
| PBDE 209 | Deca | Deca was dissolved in toluene and applied to silica gel, sand, sediment, and soil; exposed to artificial and natural sunlight for 5 to 10 days. | [86] |
| Deca degraded readily to nona- and octa-BDEs (as well as a number of other PBDEs with fewer bromines) on all matrices. A major degradation product was PBDE-183, which is a component of the now banned Octa mixture. Half of the Deca degraded within within 12 hours when applied to sand, and between 2 and 8 days when applied to sediment or soil. Shorter half-life in toluene. | |||
| 13 congeners (with 3 to 10 bromines) | Penta, Octa, Deca | Sample was dissolved in toluene and placed in either white or brown glass bottles in sunlight for 2 to 14 days. | [85] |
| Only 6% of deca-BDE remained after 5 days and only 1% after 14 days. PBDEs with 3 to 6 bromines (inlcuding the congener PBDE-47 most prevalent in wildlife and humans) found to be highly stable: they did not degrade significantly even after 14 days. | |||
| 39 congeners (with 1 to 7 bromines) | Penta, Octa | Sample was dissolved in a lipid matrix at "biologically relevant concentrations" and exposed to sunlight for 2 or 120 minutes. | [44] |
| Two of the most common congeners found in biota (PBDEs 47 and 99) were formed in significant amounts from the degradation of one hepta-, most hexa- and some penta-BDEs. |
PBDEs degrade in steps. Deca, with ten bromines, has been shown to break down into congeners with 9, 8 or 7 bromines in solvent, on silica gel, sand and soil. In one study, researchers found less than half of the original sample of Deca remaining after 20 minutes when Deca was spread on silica gel and exposed to sunlight. [86] They found that Deca persisted longest in soil — but only half remained after just 8 days. And PBDEs with 6 to 9 bromines have also been shown to break down into congeners with 5 or 4 bromines. [85] These less-brominated congeners are not only the most toxic and bioaccumulative, but also seem to be the most resistant to degradation.
It is impossible to ignore evidence of Deca breakdown. In April 2003, the EPA officially acknowledged that "higher brominated forms such as [Deca] can be altered to form more toxicologically active lower brominated forms." [43] While most research has focused on degradation in the environment, a few recent studies have added a twist: Debromination can occur in organisms themselves. [88, 83, 84] When researchers fed Deca to young fish for 60 days, almost none of it was found when the fish was tested 40 days later. But they did find seven different PBDE congeners with fewer bromines, none of which were present in the carp's diet. The researchers concluded that Deca was debrominating in the fish's tissues and converting into congeners "similar to the components in the [Penta] commercial mixtures." Even more importantly, they point out:
"Considering the high levels of [Deca] that have been measured in the environment, it is possible that over long periods of time significant formation of penta- and hexa-BDEs could occur. Therefore, banning the [Penta] commercial mixtures and continuing the use of the [Deca] mixtures may not alleviate the concerns over the presence of lower-brominated PBDEs in the environment." [84]
At this point it is hard to say how much of the Penta and other less-brominated PBDEs in people, animals and the environment originated as Deca. This is specially true given the fact that most studies looking at PBDE breakdown have been two weeks or shorter. But it is clear that allowing Deca to remain on the market may seriously undermine the environmental benefits of getting rid of Penta and Octa. To make matters worse, some data suggests that Deca can degrade into dioxin-like compounds known as PBDFs (polybrominated dibenzofurans) after exposure to ambient sunlight. [87, 89, 42.] Low levels of the very similar polychlorinated dioxins and furans are known to cause cancer and birth defects. PBDFs have recently been measured in human tissue samples and the environment in Japan. [90, 91]
How toxic are Penta and Octa PBDEs?
While research on Deca's health effects is still relatively sparse, there is a substantial body of research on the less-brominated PBDEs. Even low doses of these compounds can cause thyroid hormone disruption, permanent learning and memory impairment, behavioral changes, hearing deficits, delayed puberty onset, and decreased sperm counts in laboratory animals. Scientists have also found that exposures occurring in utero or during infancy lead to more significant harm than exposure during adulthood, and at much lower levels. Some of these studies have found toxic effects at levels lower than are now detected in American women.
Many of the known health effects of PBDEs are thought to stem from their ability to disrupt the body's thyroid hormone balance. Depressed thyroid hormone levels can cause fatigue, depression, anxiety, unexplained weight gain, hair loss and low libido in adults, and even more serious health effects in developing fetuses and infants. [92] A study of women whose levels of thyroid hormones measured in the lowest 10 percent of the population during the first trimester of pregnancy, for example, found that these women were more than 2.5 times as likely to have a child with an IQ of less than 85 (in the lowest 20 percent of the range of IQs). These women were five times as likely to have a child with an IQ of less than 70, meeting the diagnosis of "mild retardation." [93] Although no direct link could be made, one study found higher rates of hypothyroidism among workers exposed to brominated fire retardants on the job. [94]
Scientists are finding that even short-term exposures to commercial PBDE mixtures or individual congeners can alter thyroid hormone levels in animals, with effects being more profound for those animals which were exposed while they were young or in the womb. [70, 71, 72, 73, 74, 75] Other researchers have found subtle reproductive effects, such as decreased sperm count and changes in the sub-cellular structure of the ovaries, at incredibly low doses — just 0.06 mg of PBDEs per kg-day. [95] But some of the most worrisome findings come from experiments that gave young rats a low dose of PBDEs at a critical point in during their brain development. The studies found that just a single dose can cause lasting harm. [9, 11, 10]
In two different studies, one small dose of PBDEs — as little as 0.8 milligrams per kilogram of body weight per day (mg/kg-day) — given to 10-day-old mice caused "deranged spontaneous behavior," significant deficits in learning and memory and a reduced ability to adapt to new environments. These were not transient but rather "persistent neurotoxic effects" which often grew worse with age. [9, 10] Several other animal studies have shown that early-life exposures to PBDEs, often at relatively low levels, can lead to delays in sensory-motor development and hearing deficit. [11, 78, 13] Scientists still do not understand exactly how PBDEs affect neurological development. But there is evidence that they act through several different mechanisms, including mimicking thyroid hormones, increasing their rate of turnover in the body and interfering with intracellular communication. [96]
Most studies on the effects of PBDEs have been short-term — exposures of 14 days or less — so little is known about how longer term exposures may affect thyroid hormones. The answer is important, because the entire U.S. population is exposed daily to low levels of PBDEs, and studies of other thyroid hormone disrupters have found that long-term exposures can cause more serious harm at lower levels of exposure. [97] Recent studies have shown that PBDEs can act in concert with PCBs and other chemicals through similar mechanisms to increase their effects. [98, 99, 75] One study found that exposure to a combination of PCBs and PBDEs affected motor skills of lab animals ten times more strongly than exposure to either chemical alone. [9, 99]
How Safe Are Current Exposures to Penta and Octa?
Penta has been shown to cause permanent impacts to the nervous system of laboratory animals in concentrations of just 4 ppb in brain tissue and 12 ppb in brain fat. [9] One study of newborn mice exposed the animals to a single dose of PBDE-99 in a period that correlates with the third trimester of pregnancy for humans. Thirty percent of the participants in EWG's study and almost 20 percent of women in a Texas study had more than 12 ppb of PBDE-99 in the fat of their breast milk.
Studies have not yet investigated the relationship between contaminant levels measured in brain tissue with breast milk or other body tissues, making it difficult to know for sure if human exposures exceed levels known to permanently damage rodent brains. [9, 99] Scientists are most concerned about the neurological impacts of PBDEs on the fetus and young child, and there are inherent difficulties in detecting subtle impacts to learning, memory and behavior in laboratory animals or humans.
How Safe Are Current Exposures to Deca?
The European Union is completing its risk assessment for Deca, and hopes to publish a final draft sometime in 2004. Preliminary drafts took a hard look at new data on the widespread occurrence of Deca in the environment, recent research showing direct toxicity, and evidence of degradation to more toxic congeners. [39, 81] The draft acknowledges significant uncertainty in assessing Deca's safety: "The substance is persistent and the consequences of low level exposure over the lifetime of long-lived organisms cannot be predicted with any certainty from the current database." [81]
To fill these data gaps, scientists recommend continued monitoring, "for levels of both the substance and its more toxic and bioaccumulate degradation products." [81] They recommend that EU member states make decisions about Deca regulation based on principles of "precautionary risk management" rather than quantitative analysis of the data. The EU risk assessment does not include the cumulative health risks of Deca in combination with those more toxic congeners or with other chemicals such as PCBs.
PBDE Regulation
In 2003, California voted to ban Penta and Octa PBDEs by 2008, making California the first U.S state to take regulatory action. Several months later, Great Lakes Chemical, the sole American manufacturer of the two chemicals, reached a "voluntary" agreement with EPA to remove Penta and Octa from the U.S. market by 2005. In March 2004, U.S. Reps. Hilda Solis (D-CA), Lynne Woolsey (D-CA) and Diana DeGette (D-CO) introduced federal legislation to extend the voluntary phase-out and ban Penta and Octa in 2006. The bill takes another step forward by directing the EPA to identify and regulate other chemicals that break down to Penta and Octa. The bill also calls for labeling of products that contain any type of PBDE. Numerous other states have enacted or are considering studies, regulations or bans on one or more PBDEs, including Hawaii, Maine, Massachusetts, Michigan, New York and Washington state.
Overview of current legislative efforts
| State | Bill | Proposed action to regulate PBDEs |
|---|---|---|
| California | AB302 | Bans the use of Penta and Octa PBDEs by 2008. |
| Hawaii | HB 2013 | Bans the manufacture, use or sale of a product containing Penta or Octa PBDEs by 2006. |
| Massachusetts | H2275/S1268 Act for a Healthy Massachusetts | This bill will establish a pragmatic, gradual approach to reducing health impacts from ten identified toxic chemicals we are exposed to in everyday life where safer alternatives are available. Penta-PBDE is one of the chemicals listed. |
| Michigan | HB4406 (passed) | Regulates release of PBDEs (permit to release, release guidelines set by state agency). |
| HB4407 (passed) | Sets sentencing guidelines for releasing PBDEs or filing a false report. | |
| -- | Petition to Michigan Department of Environmental Quality to list PBDES as Ôcritical materialsÕ on the stateÕs list of worst chemicals. Would trigger a reporting requirement. | |
| Maine | LD1790 | Would regulate the sale of products containing brominated chemicals, requires labeling, and permits fees. |
| New York | S5712/A9207 | Prohibits manufacture or sale of products with 0.1% Penta, Octa, or Deca by mass. |
| Washington | -- | Legislature authorized $325,000 for program to phase out persistent, toxic chemicals including PBDEs. |
Because each type of fire retardant interacts differently with different materials, substituting one type of retardant for another is more complicated than it seems. But the electronics industry, in particular, has shown how quickly industry can react to the regulatory challenges. In the early 1990s, Deca and Octa were the most widely used fire retardants in the plastic housings of electronic equipment for the European market. But by 1999, PBDEs had been almost completed phased out of these plastic housings in Europe — the result of numerous individual companies choosing to move away from these toxic chemicals before their dangers had been fully mapped out. [3]
In the U.S., Intel, IBM, and Ericsson products already contain no PBDEs. Most Hewlett-Packard monitor housings are PBDE-free and the company has redesigned its computer casings so they don't require chemical fire retardants to meet safety standards. Sony, Motorola, Panasonic, NEC, Samsung and Toshiba have all taken significant steps towards phasing out chemical fire retardants from their products. [100, 101]
But there is there is little gain in replacing PBDEs with another chemical that is later found toxic. Two other brominated fire retardants with highly questionable environmental profiles, TBBPA (tetrabromobisphenol-A) and HBCD (hexabromocyclododecane), are being used as replacements for PBDEs in many products. [3] A more encouraging trend is that some companies have decided to phase out all chemicals in their products that contain chlorine or bromine.
Even better, other companies are now taking a look at the entire lifecycle of their products, reducing the use of toxins at each step in the process, or redesigning them to be more environmentally friendly. When Apple conducted a life-cycle analysis the company discovered that although using aluminum-based computer housing required more energy to produce, it also eliminated the need for chemical fire retardants, extended the product life, and allowed for easier, less toxic disposal. [101]
The bromine industry's lobbying group, the Bromine Science and Environmental Forum (BSEF), claims that "a ban of [Deca] would imply that any chemical found in [living things] or the environment should be banned no matter the level found. This would be the end of modern society as we know it." [102] BSEF's hyperbole simply doesn't stand up against the substantial progress the electronics industry has made in moving away from toxic fire retardants, and the growing evidence that Deca poses a serious environmental threat.
Beyond efforts to ban or replace a handful of brominated fire retardants, consumers will remain at risk so long as manufacturers are not required to test the effect of replacement chemicals on human health before they go on the market. There is very little data on the toxicity of the fire retardants that are currently being developed or are already in use as alternatives to PBDEs. This is largely because of well-documented shortcomings in federal toxics laws.
The chief regulatory statute for commercial chemicals, the Toxic Substances Control Act (TSCA), is infamous for its failure to lend meaningful authority to the Environmental Protection Agency. [103] The looming PBDE crisis and uncertainty surrounding replacement chemicals provide another disturbing illustration of the failures of a regulatory system that allows persistent, bioaccumulative toxins onto the marketplace before they have been adequately tested for safety. With these fire retardants, we are again reaping the high costs, in terms of health and productivity, of this industry-slanted system.
Under the current system, the EPA reviews new chemicals through a process that does not require health and safety test data and that discourages voluntary testing. Companies submit only basic toxicity data for fewer than half of all applications for new chemicals, and the government approves 80 percent of these with no use limits and no requests for tests. Eight of 10 new chemicals win approval in less than three weeks, at an average rate of seven a day. [103]
Perhaps worse than weak controls on new chemicals is the blind eye turned on the old ones. When TSCA was enacted in 1976, more than 63,000 chemicals already in use were "grandfathered" — granted blanket approval for continued use in consumer and industrial products with absolutely no requirement for further study. Most brominated fire retardants fell into this loophole and won implicit approval for widespread use in consumer products with no required health and safety testing. In 1998, the EPA and the nonprofit organization Environmental Defense reviewed all of the toxicity and environmental fate studies publicly available and found no information — not a single test — for 43 percent of the 2,600 chemicals produced in the highest volumes in the U.S. [103, 104]
The chemical industry has since agreed to do more tests to assess potential toxicity to children for a select number of the most widely use chemicals under the Voluntary Children's Chemical Exposure Program (VCCEP). The three most widely-used PBDE mixtures were included in the first group of 23 chemicals to be assessed as part of this program, but the usefulness of the VCCEP program is highly limited. Its purpose is to make "health effects, exposure, and risk information" of these chemicals available and provide "the means to understand the potential health risks to children." [105] But because the program is voluntary, chemical manufacturers are unlikely to hand over any information that might be damning for their chemical products, nor do they have much incentive to fill any significant scientific data gaps that are identified in the process.
There is no question that fire safety is important and that making products fire-resistant can save lives. Chemical fire retardants have become ubiquitous over the last few decades, but a wide variety of fire safety strategies exist. Using less-flammable materials or changing the product design so that it is inherently more fire resistant, are chemical-free solutions. Using less toxic chemicals as fire retardants is another option. We do not have to expose our children to toxins to protect them from fire.
The current system for biomonitoring in the U.S. is highly inadequate for identifying and tracking the multitude of chemicals Americans are accumulating and carrying in our bodies. The U.S. Centers for Disease Control and Prevention (CDC) have conducted two biomonitoring studies that examined up to 116 environmental chemicals in the blood and urine of 2,500 people. [106] But this is just a tiny subset of the thousands of chemicals we are exposed to every day, and will always have a lag time of several years or more when investigating news of emerging contaminants of concern such as PBDEs or any other brominated fire retardants. Furthermore, the CDC study measured only four chemicals in children younger than six years old and did not look at any contaminants in breast milk, both of which are important for estimating chemical exposures to these most sensitive subpopulations.
In an age where chemical industries are releasing millions of tons of chemicals to the environment and chemical manufacturers gain permission to put more than 2,000 new chemicals into the biosphere each year, we need a better biomonitoring system. Such a system would serve as an early warning system for chemicals that are building up in our bodies, that can track trends in chemicals levels over time, and most importantly, trigger prompt regulatory action when necessary.
Recommendations
Just as high levels of PCBs still remain in the environment today thirty years after being banned, even if all BFR production ended tomorrow, PBDEs and other brominated flame retardants will be around for many years. With 450 million pounds of PBDEs and other brominated flame retardants used each year, we must act now to limit future contamination by moving quickly to phase out these toxins and adopt more sustainable methods of fire protection.
What should government do?
- The EPA should phase out the last unregulated PBDEs. It is no longer possible to ignore evidence that Deca is a threat to human health and the environment. In the interim, all products containing brominated flame retardants must be labeled so that consumers have the option of choosing products without them.
- The EPA and Consumer Product Safety Commission must give American consumers clear advice on the safe use and disposal of products containing Penta, Octa and Deca PBDEs. Although the first two will be phased out by the end of 2004, American homes are full of products containing PBDE. Millions of mattresses and couches and miles of carpet padding and industrial drapes are sources of contamination to American homes and landfills.
- EPA must screen new and existing chemicals for their health effects. In particular, potential replacement fire retardants must be adequately tested to ensure that they are not persistent, bioaccumulative or toxic. Testing must include the outcomes most relevant to children's health. Changes in product design that decrease the need for chemical fire retardants should be encouraged over simply switching to different, less studied chemicals.
- The U.S. Centers for Disease Control and Prevention should expand the fledgling national biomonitoring program to include a greater number of chemicals and people. The study provides critical data in identifying chemicals that are accumulating in our homes, in our bodies and in the environment; tracking trends in exposure; providing data needed to more fully understand human health risks; and helping EPA and other agencies effectively transition businesses to safer, less persistent chemicals than those in current common use.
- Congress should increase funding for urgently needed additional research on toxic fire retardants, including their health effects, how they get into our homes, the human body, and current levels of accumulation in people, animals and the environment.
What should industry do?
In the absence of government regulation, U.S. manufacturers and users of chemical fire retardants should voluntarily comply with any action taken by the European Union. Chemical companies should work to minimize the toxicity of existing fire retardants and thoroughly test replacement chemicals for safety. Companies who use fire retardants in their products should follow the lead of some computer makers, who are redesigning their products so that fire retardants are not needed. Retailers should follow the example of IKEA and some other companies in demanding that their suppliers avoid the use of chemical fire retardants.
What should parents and other concerned consumers do?
EWG's studies have found that exposure to brominated fire retardants is unavoidable. EWG found them in the dust of every home and in the body of every participant tested. Our homes and offices are filled with brominated fire retardants in products including foam-padded furniture, computer and television screens, and the padding underneath our carpets.
Even if these toxic fire retardants were phased out immediately, our exposures to them would continue through the foods we eat or from the products in our households. In the absence of government safeguards to remove persistent toxins from household products, or labels which would allow consumers to choose less toxic products, parents should consider the following options:
- Avoid contact with decaying or crumbing foam that might contain fire retardants. Replace couches, stuffed chairs, automobile seats that have exposed foam. If you can not afford to replace degrading foam products, cover them with a sturdy cloth and clean up the area around them with a high efficiency vacuum (equipped with a HEPA filter.) Do not reupholster foam furniture in homes where children or pregnant women live.
- Be careful when removing and replacing the foam padding beneath carpets. Isolate the work area with plastic and avoid tracking construction dust into the rest of your home. Clean up with a HEPA-filter vacuum when you are finished.
- Buy products with natural fibers (cotton and wool) that are naturally fire resistant.

