
FDA’s advisory Science Board convened a BPA panel to evaluate an FDA staff risk assessment that termed bisphenol A (BPA) safe in food packaging. In its testimony to the panel, EWG highlighted 7 key shortcomings in the staff assessment. In every instance, the panel agreed with EWG and disagreed with the FDA staff report. The panel conclusions, released on October 28, will be formally presented to the full Science Board on October 31.
| Issue | EWG says | Advisory panel says |
| FDA estimates of infant exposure to BPA from formula | "FDA’s so called 'conservative' exposure assessment for infants significantly underestimates exposures from BPA contamination of infant formula. ...[A]nalysis of formula concentrations shows that many infants are exposed to BPA above the levels calculated by FDA." (9/12/08 pg. 10, also 9/16/08, 9/24/08)" | "The draft FDA exposure assessment...lacks an adequate number of infant formula samples and relies on mean values rather than accounting for the variability in samples." (pg. 3) "[FDA] did not adequately account for variability in potential exposures, which the invited Panel [including EWG analyst Sonya Lunder] noted could be very large." (pg. 5) |
| FDA's exclusion of studies showing effects of exposure to low doses of BPA | "FDA uses inconsistent and illogical criteria to reject [independent] studies, notably the 12 studies NTP highlights as evidence of ‘low dose’ toxicity." (9/12/08 pg. 2, also 9/16/08) | "The draft FDA report does not articulate reasonable and appropriate scientific support for the criteria applied to select data for use in the assessment." (pg. 4) |
| Differences between FDA and the National Toxicology Program | "FDA’s findings stand in contrast to conclusions of the NIH’s National Toxicology Program’s recently completed monograph assessing the developmental and reproductive effects of BPA." (9/12/08 pg. 2, also 9/16/08.) | "Consistent and credible criteria for study inclusion, recommended by the Subcommittee, would be to use those studies that are judged as “adequate” by CERHR [Center for the Evaluation of Risks to Human Reproduction, an arm of the National Toxicology Program] in the FDA hazard, dose-response and safety assessment of BPA." (pg. 4) |
| FDA's reliance on studies that are certified for "Good Laboratory Practices" | "FDA’s assessment of BPA...excludes information gathered from academic scientists who find sensitive effects to the reproductive system and behavior. (9/12/08 pg. 4, also 9/16/08) | "The Subcommittee does not agree with the exclusion of the non-GLP studies in the safety assessment." (pg. 6) |
| The availability of new evidence | "FDA did not review very recent studies finding brain effects in primates and linking BPA exposures in American adults to heart disease and diabetes." (10/24/08 pg. 2) | "Several studies of effects of BPA on adult humans and animal species that were published after the draft assessment was finished should be considered for inclusion in the final assessment." (pg. 7 & 12) |
| FDA's determination of a "No Effect Level" at 5 mg per kilogram per day | "FDA should use the lowest dose toxicity studies to calculate risk. Particularly the number of studies finding permanent effects to brain and behavior and the male reproductive system at 10 ug/kg-bodyweight per day." 9/16/08, 10/24/08 pg. 2) | "The weight-of-the-evidence...provides scientific support for use of a point of departure substantially below (i.e., at least one or more orders of magnitude lower than) the 5 mg/kg bw/day level selected in the draft FDA assessment." (pg. 4) |
| FDA's statement that infant's exposures are 2,000 times lower than the "No Effect Level" and therefore safe | "FDA has failed to acheive [its] safety standard." and "Using the low dose studies recommended by expert advisors as a “point of departure” would decrease the margin of safety by a factor of 500, meaning that FDA’s estimated exposures for a formula-fed baby would be just 4 times less than the doses that cause permanent brain and behavior impairment in laboratory tests." (10/24/08 pg. 2) | "Coupling together the available qualitative and quantitative information...provides a sufficient scientific basis to conclude that the Margins of Safety defined by FDA as “adequate” are, in fact, inadequate." (pg. 4) |
FDA’s has failed to support its claim that current uses of BPA are safe. FDA must start from the ground up with a new assessment that accurately characterizes BPA risks for infants, children and adults. In the meantime FDA must take immediate actions to reduce infant exposures to this toxic chemical. At a minimum this would involve Canadian-type precautions to reduce immediately infant exposures from infant formula and food containers. Formula-makers can and should reduce BPA levels while safer packaging is investigated. Parents should be advised to use non-polycarbonate bottles and powdered formula wherever possible. These dramatic measures are needed to assure that infants are not exposed to harmful amounts of BPA while a health-protective safety assessment is being drafted.
EWG Comments to BPA Subcommittee on the Risks of Liquid Formula
Download PDF file of this letter.
September 24, 2008
Martin Philbert, PhD
BPA Subcommittee Chair
c/o Office of Science and Health Coordination
Office of the Commissioner (HF-33)
Food and Drug Administration
Rockville, MD 20857
Re: Canadian study and exposure calculations for BPA in formula
Dear Dr. Philbert:
We would like to submit to the sub-Committee a recent publication of BPA levels in Canadian infant formula (Cao 2008, attached). These tests include 21 liquid formula types purchased in 2007. Four formula companies—Abbott Nutrition, Mead-Johnson, PBM and Nestlé—dominate the U.S. and Canadian market, so the results would be reasonably reflective of exposures for American infants. These tests underscore the need for examining high-end BPA exposures for babies. Health Canada’s exposure assessment addresses 2 of the 4 gaps identified by the sub-committee and the panel, and results in a tripling of the exposure estimate for hungry children fed from the worst brands.
I’d like to note a couple of key findings from the Canadian government’s tests:
Eighty percent (80%) of the formulas tested exceed the BPA concentration used by FDA in their draft assessment. FDA’s exposure estimate uses an average BPA level of 2.5 ppb in canned formula, and 17 of 21 formula-types exceed this value. Three ready to eat formulas were tested, but only 1 was for infants younger than 12 months.
The tests also suggest differences between brands. One company had consistently elevated levels compared to the others. The authors postulate that it could be due to the thickness of the epoxy lining used in company cans. The chemists tested at least 2 separate cans of each formula type and found high concordance among duplicate samples with an average difference of just 7.5%. Brand variability and can consistency underscores the need to calculate exposure based on the high-end formula measurements, assuming that a child is fed the same brand of formula throughout infancy.
Health Canada estimates high-end exposures by including maximum ingestion volumes and maximum BPA measurements in their calculations. These steps result in an intake estimate for BPA from canned formula that is 3 times higher than FDA's estimate over the first 7 months of life. Exposures >1 ug/kg-day from formula alone could continue through the entire first 7 months of infancy for hungrier infants, those fed ready-toeat formula, or those whose parents choose the brand with the greatest BPA concentrations.
Health Canada’s high-end exposure estimates for infants are 3 times FDA’s estimate. (ug/kg-day)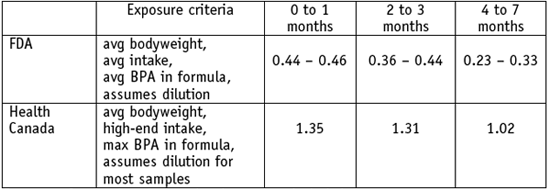
Exposures from polycarbonate bottles are not included Health Canada’s estimates do not estimate increased intensity of exposure for lighter babies, nor do they fully explore risks for those fed undiluted or ‘ready-to-eat’ formula. FDA should also include these 2 exposure factors in their analysis of highly exposed infants.
The authors did not detect BPA in any of the 56 canned powdered formulas tested (with a detection limit of 1 ppb), suggesting that safer alternatives exist for most infants. EWG shares the concerns raised by panelists about infants who require liquid formulations (due tap water quality or medical concerns). In our opinion the risks posed by BPA ingestion during infancy indicate that parents should select powdered formula or liquid formulas packaged in non-metal containers while safer can linings are developed.
Sincerely,
Sonya Lunder, MPH
Senior Analyst
Environmental Working Group
Attached:
Cao XL, Dufresne G, Belisle S, Clement G, Falicki M, Beraldin F, Rulibikiye A. 2008. Levels of bisphenol A in canned liquid infant formula products in Canada and dietary intake estimates. J Agric Food Chem. Sep 10;56(17):7919-24. Epub Aug 15 2008.
EWG comments on BPA assessment to full Science Board
Download PDF file of this letter.
October 24, 2008
Barbara J. McNeil, M.D., Ph.D.,
Science Board Chair
c/o Office of Science and Health Coordination
Office of the Commissioner (HF-33)
Food and Drug Administration
Rockville, MD 20857
Re: Science Board meeting on Bisphenol A toxicity
Dear Dr. McNeil,
We are dismayed that the Science Board has chosen not to delay its BPA meeting despite allegations of serious conflict of interest regarding donations to the subcommittee chair Dr. Martin Philbert. EWG, Congressional leaders and ethicists have called for a full investigation of financial conflicts of subcommittee members and any role these undisclosed donations played in the subcommittee’s determination on BPA safety. However the rush to make a final judgment without a full investigation of undisclosed ties to affected parties will cast a cloud on any determination of BPA safety.
Environmental Working Group has repeatedly raised concerns about significant flaws in FDA’s draft risk assessment for BPA in food packaging, both in advance of the September 16, 2008 subcommittee meeting, as well as in a follow-up letter to Dr. Philbert and the subcommittee on September 24th. Both documents are attached.
Here are our key concerns:
FDA estimates that infant exposures are 2,000 times lower than the levels toxic to animals. But this margin of safety is seriously eroded by two factors. FDA seriously underestimates BPA exposures for infants who ingest the most BPA from formula. Furthermore, FDA ignored dozens of studies indicating BPA toxicity at incredibly low doses. These failures put FDA out of step with the conclusions of the National Toxicology Program and the Canadian government.
- FDA used an unacceptably low estimate of BPA levels in formula. Eighty percent (80%) of 21 liquid formulas tested by Health Canada had more BPA than the concentration (2.5 ppb) used in FDA’s risk calculations (Cao 2008). This leads to an underestimate of infant exposure to BPA from formula by a factor of 3. FDA also fails to account for the risks for lighter and hungrier babies. Finally, risks to premature babies, who are commonly fed liquid formula, are not assessed. Correcting these reckless assumptions will erode FDA’s ostensible 2,000-fold margin of safety for infants.
- FDA bases its safety assessment on widely criticized industry-funded studies that are insensitive to low dose effects of BPA. This issue was widely discussed at the subcommittee meeting, both in presentations by the NTP and by expert advisors to the subcommittee. The studies are in a recent issue of Environmental Health Perspectives (Myers 2008).
- FDA’s decision to ignore key studies showing brain and behavioral impacts of BPA during development was based on an ACC-funded literature review that came to dramatically different conclusions than those of Canada’s and the NTP (FDA 2008).
- Based on evidence of low-dose toxicity, four of seven expert advisors who briefed the BPA subcommittee at its September meeting recommended that FDA consider a dose level of 10 ug/kg-day as a “point of departure” for its risk assessment.
- FDA did not review very recent studies finding brain effects in primates (Leranth 2008) and linking BPA exposures in American adults to heart disease and diabetes (Lang 2008).
These shortcomings have a major impact on the proposed safety assessment for BPA. Using the low dose studies recommended by expert advisors as a “point of departure” would decrease the margin of safety by a factor of 500, meaning that FDA’s estimated exposures for a formula-fed baby would be just four times less than the doses that cause permanent brain and behavior impairment in laboratory tests. This would suggest that current exposures are clearly unacceptable, given FDA’s goal of at least a 1000-fold difference between human exposures and doses that cause toxic effects (FDA 2008). Adjusting infant intake estimates to include highly exposed infants would also result in a safety margin of less than 1,000.
FDA’s draft safety standard has failed to support its claim that current uses of BPA are safe. FDA defines safety in 21 CFR §170.3(i) as, “reasonable certainty in the minds of competent scientists that the substance is not harmful under the intended conditions of use.” (FDA 2008) The concerns voiced by NIH’s National Toxicology Program (NTP) over the safety of current BPA exposures for the fetus, infant and young child, (NTP 2008) and conclusions of the 38 independent scientists participating in the recent NIH-sponsored review of BPA toxicity are sufficient evidence of dissent by competent scientists. The 38 scientists caution:
“[E]vidence suggests that these adverse effects are occurring in animals within the range of exposure to BPA of the typical human living in a developed country, where virtually everyone is exposed to measurable blood, tissue and urine levels of BPA that exceed the levels produced by doses used in the “low dose” animal experiments.” (vom Saal 2007)
FDA’s conclusion also stands in clear contrast to the recent action by the Canadian government (Environment Canada 2008a,b). Canada determined that low dose BPA brain and behavior studies and the intensity of infant exposures demonstrate that low dose BPA presents an unacceptable risk of harm. Canadian health officials also decided that immediate action was warranted to reduce infants’ ingestion of BPA. On October 18, 2008, Canada announced a ban polycarbonate bottles and steps to reduce BPA in liquid formula via a “minimum reasonably achievable” standard (Environment Canada 2008b).
The Canadian formula action will dramatically reduce BPA exposures for highly exposed infants. Canadian tests found significant variability in BPA levels by brand: the worst formula brands have on average 4 times more BPA than the best brands (Cao 2008). Canada found no detectable BPA in powdered formula, despite the fact that some formula-makers use it in the lids of powdered containers. Canada’s actions to reduce BPA levels in formula may trickle into the U.S., since the same companies produce formula for both countries.
Given the numerous technical flaws in the FDA document, new evidence of harm, and unaddressed concerns about conflict of interest, we must recommend that the Science Board stop the current review and send FDA back to the drawing board to produce an assessment that accurately represents BPA’s risks to children and adults.
However, since BPA-free bottles and powdered formula are widely available, FDA should at a minimum enact Canadian-type precautions to reduce immediately infant exposures from infant formula and food containers. Formula-makers can and should reduce BPA levels while safer packaging is investigated. Parents should be advised to use non-polycarbonate bottles and powdered formula wherever possible. These dramatic measures are needed to assure that infants are not exposed to harmful amounts of BPA while a health-protective safety assessment is being drafted.
Sincerely,
Sonya Lunder MPH
Environmental Working Group
1436 U St, NW, Suite 100
Washington, DC 20009
Cc: Frank M. Torti, M.D., M.P.H.,
FDA principal deputy commissioner
Andrew von Eschenbach, FDA Commissioner
Attached: EWG comments to BPA subcommittee, Sept. 12, 2008. Available online: https://static.ewg.org/files/BPA_091208.pdf EWG comments to BPA subcommittee, Sept. 24, 2009.
References:
Cao XL, Dufresne G, Belisle S, Clement G, Falicki M, Beraldin F, Rulibikiye A. 2008. Levels of bisphenol A in canned liquid infant formula products in Canada and dietary intake estimates. J Agric Food Chem. Sep 10;56(17):7919-24. Epub Aug 15 2008.
Environment Canada. 2008a. Screening Assessment for The Challenge Phenol, 4,4' - (1-methylethylidene)bis- (Bisphenol A). Chemical Abstracts Service Registry Number 80-05-7. Available: http://www.ec.gc.ca/substances/ese/eng/challenge/batch2/batch2_80-05-7.cfm [accessed Oct. 22 2008].
Environment Canada. 2008b. Proposed Risk Management Approach for Phenol, 4,4' - (1-methylethylidene)bis- (Bisphenol A). Chemical Abstracts Service Registry Number 80-05-7. Available: http://www.ec.gc.ca/substances/ese/eng/challenge/batch2/batch2_80-05-7_rm.cfm [accessed Oct. 22 2008].
Food and Drug Administration (FDA). 2008. Draft assessment of bisphenol A for usein food contact application. Available: http://www.fda.gov/ohrms/dockets/AC/08/briefing/2008-0038b1_01_00_index.htm, downloaded 9/09/08
Lang IA, Galloway TS, Scarlett A, Henley WE, Depledge M, Wallace RB, Melzer D. 2008. Association of urinary bisphenol A concentration with medical disorders and laboratory abnormalities in adults. JAMA. 2008 Sep 17;300(11):1303-10. Epub 2008 Sep 16.
Leranth C, Hajszan T, Szigeti-Buck K, Bober J, MacLusky NJ. 2008. Bisphenol A prevents the synaptogenic response to estradiol in hippocampus and prefrontal cortex of ovariectomized nonhuman primates. Proc Natl Acad Sci U S A. 2008 Sep 16;105(37):14187-91. Epub 2008 Sep 3.
Myers JP, et al. 2008. Why Public Health Agencies Cannot Depend upon ‘Good Laboratory Practices’ as a Criterion for Selecting Data: The Case of Bisphenol A. Environmental Health Perspectives. In press.
vom Saal FS, Akingbemi BT, Belcher SM, Birnbaum LS, Crain DA, Eriksen M, et al. 2007. Chapel Hill bisphenol A expert panel consensus statement: integration of mechanisms, effects in animals and potential to impact human health at current levels of exposure. Reprod Toxicol 24(2):131-138.

