
EWG finds heated Teflon pans can turn toxic faster than DuPont claims
 In two to five minutes on a conventional stovetop, cookware coated with Teflon and other non-stick surfaces can exceed temperatures at which the coating breaks apart and emits toxic particles and gases linked to hundreds, perhaps thousands, of pet bird deaths and an unknown number of human illnesses each year, according to tests commissioned by Environmental Working Group (EWG).
In two to five minutes on a conventional stovetop, cookware coated with Teflon and other non-stick surfaces can exceed temperatures at which the coating breaks apart and emits toxic particles and gases linked to hundreds, perhaps thousands, of pet bird deaths and an unknown number of human illnesses each year, according to tests commissioned by Environmental Working Group (EWG).
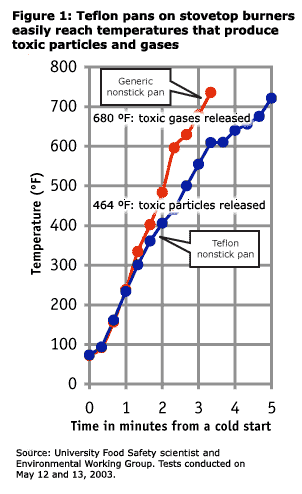
In new tests conducted by a university food safety professor, a generic non-stick frying pan preheated on a conventional, electric stovetop burner reached 736°F in three minutes and 20 seconds, with temperatures still rising when the tests were terminated. A Teflon pan reached 721°F in just five minutes under the same test conditions (See Figure 1), as measured by a commercially available infrared thermometer. DuPont studies show that the Teflon offgases toxic particulates at 464°F. At 680°F Teflon pans release at least six toxic gases, including two carcinogens, two global pollutants, and MFA, a chemical lethal to humans at low doses. At temperatures that DuPont scientists claim are reached on stovetop drip pans (1000°F), non-stick coatings break down to a chemical warfare agent known as PFIB, and a chemical analog of the WWII nerve gas phosgene.
For the past fifty years DuPont has claimed that their Teflon coatings do not emit hazardous chemicals through normal use. In a recent press release, DuPont wrote that "significant decomposition of the coating will occur only when temperatures exceed about 660 degrees F (340 degrees C). These temperatures alone are well above the normal cooking range."
These new tests show that cookware exceeds these temperatures and turns toxic through the common act of preheating a pan, on a burner set on high.
In cases of "Teflon toxicosis," as the bird poisonings are called, the lungs of exposed birds hemorrhage and fill with fluid, leading to suffocation. DuPont acknowledges that the fumes can also sicken people, a condition called "polymer fume fever." DuPont has never studied the incidence of the fever among users of the billions of non-stick pots and pans sold around the world. Neither has the company studied the long-term effects from the sickness, or the extent to which Teflon exposures lead to human illnesses believed erroneously to be the common flu.
The government has not assessed the safety of non-stick cookware. According to a Food and Drug Administration (FDA) food safety scientist: "You won't find a regulation anywhere on the books that specifically addresses cookwares," although the FDA approved Teflon for contact with food in 1960 based on a food frying study that found higher levels of Teflon chemicals in hamburger cooked on heat-aged and old pans. At the time, FDA judged these levels to be of little health significance.
Of the 6.9 million bird-owning households in the US that claim an estimated 19 million pet birds, many don't know know that Teflon poses an acute hazard to birds. Most non-stick cookware carries no warning label. DuPont publicly acknowledges that Teflon can kill birds, but the company-produced public service brochure on bird safety discusses the hazards of ceiling fans, mirrors, toilets, and cats before mentioning the dangers of Teflon fumes.
As a result of the new data showing that non-stick surfaces reach toxic temperatures in a matter of minutes, EWG has petitioned the Consumer Product Safety Commission (CPSC) to require that cookware and heated appliances bearing non-stick coatings must carry a label warning of the acute hazard the coating poses to pet birds. Additionally, we recommend that bird owners completely avoid cookware and heated appliances with non-stick coatings. Alternative cookware includes stainless steel and cast iron, neither of which offgases persistent pollutants that kill birds.
EWG Petition to the CPSC (PDF).
Teflon kills birds
Avian veterinarians have known for decades that Teflon-coated and other non-stick cookware can produce fumes that are highly toxic to birds. As early as 1986, a Chicago-area expert on “Teflon toxicosis” called the phenomenon a “leading cause of death among birds,” and estimated that hundreds of birds are killed by the fumes and particles emitted from Teflon-coated products each year [1][2]. Although an accurate national accounting of deaths is not available, in a single year this Chicago veterinarian documented 296 bird deaths in 105 cases involving non-stick cookware.
Under ordinary cooking scenarios, Teflon kills birds. A review of the literature and bird owners’ accounts of personal experience with Teflon toxicosis shows that Teflon can be lethal at normal cooking temperatures, with no human lapses in judgment or wakefulness.
Bird deaths have been documented during or immediately after the following normal cooking scenarios:
- New Teflon-lined Amana oven was used to bake biscuits at 325°F; all the owner’s baby parrots died [3] [4].
- Four stovetop burners, underlined with Teflon-coated drip pans, were preheated in preparation for Thanksgiving dinner; 14 birds died within 15 minutes [2] [5].
- Nonstick cookie sheet was placed under oven broiler to catch the drippings; 107 chicks died [2].
- Self-cleaning feature on the oven was used; a $2,000 bird died [5].
- Set of Teflon pans, including egg poaching pan, were attributed to seven bird deaths over seven years [6].
- Water burned off a hot pan; more than 55 birds died [7].
- Electric skillet at 300°F and space heater were used simultaneously; pet bird died [8].
- Toaster oven with a non-stick coating was used to prepare food at a normal temperature; bird survived but suffered respiratory distress [9].
- Water being heated for hot cocoa boiled off completely; pet bird died [10].
- Grill plate on gas stove used to prepare food at normal temperatures; two birds died on two separate occasions [11].
DuPont claims that its coating remains intact indefinitely at 500°F [12]. Experiences of consumers whose birds have died from fumes generated at lower temperatures show that this is not the case. In one case researchers at the University of Missouri documented the death of about 1,000 broiler chicks exposed to offgas products from coated heat lamps at 396°F [13].
DuPont also claims that human illness will be produced only in cases involved gross overheating, or burning the food to an inedible state [12]. Yet DuPont's own scientists have concluded that polymer fume fever in humans is possible at 662°F, a temperature easily exceeded when a pan is preheated on a burner or placed beneath a broiler, or in a self-cleaning oven [14].
References
[1] Dale, Steve. 1995. “Fatal fumes; while people may not be in danger, the kitchen is no place for pet birds.” Chicago Tribune. March 26, 1995.
[2] Daniels, Mary. 1987. “Health debate; non-stick drip pans catch heat.” Chicago Tribune. March 29 1987.
[3] Stewart Bob. 2002. Personal communication with Dr. Jennifer Klein, Environmental Working Group. May 9, 2002.
[4] Stewart Bob. 2002. Personal email communication with Anne Morgan, Environmental Working Group. [date]
[5] Daniels, Mary. 1986. “Stove fumes killing cages birds; overheating coated pans can bring quick death,” Chicago Tribune. March 9, 1986.
[6] Hopkins, Steve 2001. “Bird deaths linked to Teflon coating.” Waikato Times. Hamilton, New Zealand. Independent Publishers Ltd. July 11, 2001. Copyright 2001 Independent Publishers Ltd.
[7] Kreger Theresa 2003. "Teflon deaths." Email correspondence to EWG. April 2003.
[8] Shively Carol. 2003. "PTFE fumes kill family's pet birds!" Accessed online at www.quakerville.com/qic/ezine/96Issue5/qteflon.htm. April 2003.
[9] Grahme 2003. "Teflon-related bird information." Email correspondence to Environmental Working Group. April 24 2003.
[10] Anonymous 2003. Email correspondence to Environmental Working Group. April 2003.
[11] Anonymous 2003. Email correspondence to Environmental Working Group. April 2003.
[12] DuPont 2003a. "Consumer products help: Cookware safety. Will cooking fumes generated while cooking with non-stick cookware harm people or animals, especially pet birds?" Accessed online May 10 2003 from http:/www.teflon.com.
[13] Boucher M, Ehmler TJ, Bermudez AJ. 2000. Polytetrafluoroethylene gas intoxication in broiler chickens. Avian Dis 44:449-53.
[14] Waritz, R.S. 1975. An industrial approach to evaluation of pyrolysis and combustion hazards. Environ Health Perspect 11:197-202.
An agonizing death
In cases of “Teflon toxicosis,” as the bird poisonings are called, the lungs of exposed birds hemorrhage and fill with fluid, leading to suffocation. In a laboratory study in which birds were killed by non-stick fumes, Dr. Roger Wells, a veterinarian at Michigan State University, characterized the response in many of the exposed birds as “acute respiratory distress and rapid death” [1]. Wells' description of the progression of death in a group of birds exposed to PTFE fumes is consistent with most other documented cases of Teflon toxicosis, in which birds suffer for minutes or even hours before death:
“Clinical signs of toxicosis were varied and began with eyelid blinking at 8 to 10 minutes of exposure and progressed to open-beak panting, biting at the wire cage, incoordination, wing stretching and flapping, and chirping, and usually ended in a terminal convulsion, with the birds recumbent on their sides or backs.”
The birds in this exposure group died within 17 to 27 minutes of exposure. In the same study birds in a lower dose group died up to 11 hours after exposure. The observations of pet bird owners who have lost birds to Teflon toxicosis are consistent with Wells' descriptions. Bird owners have described their birds gasping for breath, or laboring for breath with head bobbing back and forth before death. | View Bird death diaries
Scientists who have studied the lungs of birds killed by Teflon fumes find common signs of gross lung damage, including severe lesions in the most sensitive birds. Hallmark signs of PTFE poisoning in birds are lung congestion, fluid accumulation of the lung (edema), bleeding in the lung (hemorrhage), heart tissue death (necrosis of the atrial epithelium) and crystalline particulate deposition in certain lung cells (parenchyma). Autopsies also show brain hemorrhage, liver congestion, degeneration and necrosis; and heart muscle (myocardial) degeneration and necrosis.
Dr. Larry Thompson, a resident in veterinary toxicology at the University of Illinois College of Veterinary Medicine at Champaign-Urbana, says birds affected by Teflon toxicosis "show acute respiratory distress, dyspnea or difficulty breathing; they go into little rocking movements, ataxia; they look asleep or somnolent; then they go into agonal convulsions" [2]. In another account, the progression of death is described: “...the birds’ lungs are scorched, causing blood vessels to rupture, and, within minutes, the poor animals literally suffocate in their own blood.” [3].
References
[1] Wells RE, Slocombe RF, Trapp AL. 1982. Acute toxicosis of budgerigars (Melopsittacus undulatus) caused by pyrolysis products from heated polytetrafluoroethylene: clinical study. Am J Vet Res 43:1238-42.
[2] Daniels, Mary. 1986. “Stove fumes killing cages birds; overheating coated pans can bring quick death,” Chicago Tribune. March 9, 1986.
[3] Dale, Steve. 1995. “Fatal fumes; while people may not be in danger, the kitchen is no place for pet birds.” Chicago Tribune. March 26, 1995.
Few birds are saved
Very few birds survive once observable symptoms of Teflon toxicosis set in. According to Dr. Ken Welle, assistant professor of avian and exotic medicine at the University of Illinois at Champaign-Urbana, "We have saved birds by administering immediate application of steroids, but most never make it as far as our office" [1].
References
[1] Dale, Steve. 1995. “Fatal fumes; while people may not be in danger, the kitchen is no place for pet birds.” Chicago Tribune. March 26, 1995.
DuPont has known for 50 years
Environmental Working Group reviewed 16 peer-reviewed studies detailing experiments conducted over the past 50 years, showing that heated Teflon decomposes to 15 types of toxic gases and particles. Many of these studies were conducted by DuPont’s own scientists, who began studying heated Teflon (PTFE) in the 1950s when DuPont workers were developing polymer fume fever that the company found could lead to a potentially fatal condition called pulmonary edema [1]. Since DuPont's discovery of polymer fume fever, cases have been reported in the peer-reviewed literature of the same illness stemming from home kitchen exposures [2, 3].
Teflon offgasing studies show that at the design temperatures of conventional kitchen appliances, Teflon chemicals break apart to form the following particulates and gases:
- Two chemicals linked to cancer or tumors in laboratory studies (PFOA and TFE);
- Two chemicals that are potent global warming gases (PFB and CF4);
- Two chemical warfare agents (PFIB and MFA) and a chemical analog of WWII nerve gas phosgene (COF2);
- At least two chemicals that have widely contaminated the world (PFOA and TFA), one currently undergoing a rigorous safety review at the Environmental Protection Agency (PFOA);
- Four gaseous chemicals and some components of the particulate matter that are highly persistent environmental pollutants, that likely never break down in the environment (TFA, PFOA, CF4, PFB, and the perfluorinated particulate alkanes); and
- Four chemicals that are considered highly toxic relative to most other industrial chemicals (PFIB, MFA, COF2, HF).
Studies show that the gases that come off of non-stick pans are complex mixtures that vary in composition with temperature. At any given temperature the gas comprises one or more dominant chemicals, and other chemicals present in trace quantities. In numerous studies scientists have studied mortality in rats and birds exposed to the offgas mixtures, but potential long-term health impacts have not been studied. The government has not conducted a safety study of Teflon cookware. Accumulation of the offgas chemicals in food has not been studied. The potential effects to humans of inhalation exposures have not been studied, but several of the offgas components are considered highly toxic to humans relative to most other industrial chemicals.
DuPont scientists list the hallmark human symptoms of polymer fume fever as tightness of chest, malaise, shortness of breath, headache, cough, chills, temperatures between 100 and 104°F, and sore throat, based on a survey of complaints registered by workers who were struck by the illness [1]. Based on this suite of symptoms, cases of polymer fume fever from home exposures could easily be mistaken for the common flu.
The toxic particles and gases identified as Teflon offgas products, and the temperature at which they are first identified in the studies reviewed, are shown below, with toxicity information that is drawn primarily from high dose animal studies, the only source of information available for most of the chemicals:
1. 464°F - Ultrafine particulate matter [4]: Teflon produces very small (ultrafine) particles which are very toxic, causing extreme lung damage to rats within 10 minutes of exposure. Longer exposures cause death. At higher temperatures, Teflon also produces toxic gases. Some scientists have found that the particles and gases together are responsible for Teflon's toxicity, perhaps because the gases adsorb to the particles, which because of their small size can lodge deep in the lower respiratory tract [5].
2. 680°F - Tetrafluoroethylene (TFE) [6]: The National Toxicology Program considers tetrafluoroethylene (TFE) to be a “reasonably anticipated” human carcinogen because it is known to cause cancer in laboratory animals, but has not been adequately studied in people. In rats, inhaled TFE causes tumors of the kidney tubules, liver, blood vessels in the liver and one form of leukemia (mononuclear). Mice that breath TFE develop tumors of the liver and tumors that develop in blood vessels in the liver or white blood cells [7].
3. 680°F - Hexafluoropropene (HFP) [6]: In people, air exposure to fluorocarbons like HFP can lead to eye, nose and throat irritation; heart palpitations, irregular heart rate, headaches, light-headedness, fluid accumulation in the lung (edema) and possibly death. Long-term exposure in workers is associated with decreased motor speed, memory and learning [8].
In mice and rats, inhalation of hexafluoropropene (HFP) causes kidney lesions, decreased numbers of a type of immune cell (lymphocyte) and increased urination [9]. HFP also causes increased numbers of chromosomal abnormalities in hamster ovaries [8].
HFP can also be added to pesticides as an “inert” ingredient [10], which does not mean that it is non-toxic, but only that is not the pesticide active ingredient. Another example of a pesticide inert ingredient is butyl benzyl phthalate, a chemical well known to cause serious birth defects of the male reproductive system in laboratory animals.
4. 680°F - Trifluoroacetic acid (TFA) [6]: Very few studies have looked at the toxicity of trifluoroacetic acid (TFA), but those that have found decreased growth of fetal rat bone-forming cells (osteoblast) and cartilage cells (chondrocytes) [11], and neural tube defects in rat embryos at high concentrations [12]. Other studies show that HCFC-123, a hydrofluorocarbon that breaks down into TFA, causes enlarged liver and decreased levels of glucose, triglyceride and cholesterol in adult animals. But, it is unclear whether these effects are due to HCFC-123 or a metabolite [13]. A monkey study found the TFA concentration in the fetus was two to six times higher than in the mother’s blood following dosing with HCFC-123 [14].
The long-term environmental impacts of TFA are unknown, but it is extremely persistent and toxic to plants. TFA is also a breakdown product of many hydrochlorofluorocarbon (HCFCs) and hydrofluorocarbons (HFCs) used as replacement for chlorofluorocarbons (CFCs), which are potent ozone depleters used in refrigeration systems, aerosols and other products. Recently, scientists have suggested that high levels of TFA in the environment could be partly due to heated Teflon and other fluoropolymers because measured environmental levels are higher than predicted, based on breakdown of HCFCs and HFCs alone [6].
5. 680°F - Difluoroacetic acid (DFA) [6]: Very little is known about the toxicity of difluoroacetic acid (DFA), although kidney toxicity has been reported in rats [15].
6. 680°F - Monofluoroacetic acid (MFA, fluoroacetic acid or compound 1080) [6]: Monofluoroacetic acid is extremely toxic, doses as low as 0.7 to 2.1 mg/kg can kill people [16]. Initially, people report nausea, vomiting, numbness, tingling, anxiety, muscle twitching, low blood pressure and blurred vision. If exposure is high enough, people can have irregular heart rate (ventricular fibrillation), heart attacks, and severe convulsions leading to respiratory failure [17].
MFA quickly breaks down into a chemical called fluoroacetate. Sodium fluoroacetate was previously used as a powerful rodent killer (rodenticide). In the body, it breaks down into sodium and fluoroacetate, which is responsible for the toxicity. Sodium fluoroacetate kills rodents, and other animals, by inhibiting the tricarboxylic acid (TCA) cycle which transforms energy found in food to energy the body uses. Sodium fluoroacetate also causes heart and respiratory failure, central nervous system depression and damage to the testes, including decreased sperm production [18].
7. 680°F - Perfluorooctanoic acid (PFOA) [6]: Perfluorooctanoic acid (PFOA) has recently come under significant EPA scrutiny. According to Stephen L. Johnson, Assistant Administrator of EPA's Office of Prevention, Pesticides, and Toxic Substances, the EPA" will be conducting its most extensive scientific assessment ever undertaken on this type of chemical". EPA is concerned about PFOA because it never breaks down in the environment, is found in the blood of over 92 percent of Americans, and is very toxic to rats and monkeys. PFOA causes four types of tumors in rats: liver, pancreas, mammary gland (breast) and testes. PFOA also decreases thyroid hormone levels, a known risk factor for impaired brain development, and delays sexual maturation in laboratory animals. PFOA is especially toxic to the young because it kills young rats at doses that do not kill parental animals. Industry scientists estimate that it takes 4.4 years for people to eliminate just half the amount of PFOA found in their bodies. EPA is taking a close look at PFOA because levels found in the blood of people are too close to levels in rat blood that harm the animals [19, 20].
8. 878°F - Silicon tetrafluoride (SiF4) [21]: Silicon tetrafluoride is a highly toxic, corrosive gas. In the lungs, moisture causes the silicon particles to separate, releasing toxic hydrofluoric acid and also coating the lung with silicon particles. Inhaling hydrofluoric acid can cause eye and throat irritation, cough, difficult breathing, bluish skin color caused by lack of oxygen, lung damage and fluid accumulation in the lung (edema). Long term exposure can cause weight loss, decreased numbers of red and white blood cells (anemia and leukopenia), discoloration of the teeth and abnormal thickening of the bone (osteosclerosis) [22]
9. 887°F - Perfluoroisobutene (PFIB) [23]: Perfluoroisobutene (PFIB) is extremely toxic and inhalation can lead to fluid build up in the lung (edema), a condition that can lead to death. PFIB is listed in the Chemical Weapons Convention as a Schedule 2 compound [24]. PFIB is about ten times more toxic than phosgene, a highly toxic corrosive gas also listed as a chemical weapon. In water, PFIB breaks down into hydrogen fluoride which is also very toxic (see below). Short-term symptoms of PFIB exposure in people include bad taste in mouth, nausea and weakness. Lung edema occurs about one to four hours after exposure, which is life-threatening in some cases, but in most people clears up in about 3 days [25].
10. 932°F - Carbonyl fluoride (COF2) [26]: Breakdown of Teflon (PTFE) in air is the major source of carbonyl fluoride exposure [27]. Carbonyl fluoride is the fluorine version of phosgene, a chlorinated chemical warfare agent. Carbonyl fluoride fumes can irritate eyes, ears and nose. More serious symptoms of exposure include chest pains, breathing difficulty, fluid accumulation in the lungs, weakness, liver damage and increased glucose levels. Because carbonyl fluoride breaks down into hydrogen fluoride and carbon dioxide, it causes many of the same toxic effects as hydrogen fluoride (see below) [27].
11. 932°F - Hydrogen fluoride (HF) [26]: Hydrogen fluoride (HF) is a toxic corrosive gas, and can cause death to any tissue it comes into contact with, including the lungs. The toxicity of HF is due to the fluoride ion and not the hydrogen ion. Breathing HF can cause severe lung damage, such as fluid buildup in the lungs (edema) and inflammation of lung passages (pneumonia) [28].
The fluoride ion (charged particle) is extremely toxic. It is a small ion and weak acid that diffuses quickly and can pass through tissues with relative ease. Fluoride ions inhibit cell respiration, decreasing production of ATP, the major form of chemical energy used by the body. Fluoride attracts cell membranes causing cells to die. The fluoride ion is negatively charged and naturally likes to react with positively charged ions in the body like calcium and magnesium. When fluoride and calcium bind, creating a “precipitate,” a life-threatening condition of decreased calcium (hypocalcemia) can occur. Left untreated, decreases in calcium (and magnesium) can cause abnormal heart rhythm leading to heart attack, muscle spasms and death. Calcium administration is the main treatment for HF poisoning [28].
12. 1112°F - Trifluoroacetic acid fluoride (CF3COF) [21]: Trifluoroacetic acid fluoride is toxic, mostly because it breaks down into hydrogen fluoride, which is very toxic, and trifluoroacetic acid.
The few studies that have looked at the toxicity of TFA found decreased growth of fetal rat bone-forming cells (osteoblast) and cartilage cells (chondrocytes) [11], and neural tube defects in rat embryos at high concentrations [12]. Other studies show that HCFC-123, a hydrofluorocarbon that breaks down into TFA, causes enlarged liver and decreased levels of glucose, triglyceride and cholesterol in adult animals, but it is unclear whether these effects are due to HCFC-123 or a metabolite [13]. A monkey study found TFA in the fetus was two to six times higher than in the mother’s blood following dosing with HCFC-123, a hydrofluorocarbon that breaks down into TFA [14].
Fluoride ion (charged particle) is extremely toxic. It is a small ion and weak acid that diffuses quickly and can pass through tissues with relative ease. Fluoride ions inhibit cell respiration, decreasing production of ATP, the major form of chemical energy used by the body. Fluoride attracts cell membranes causing cells to die. The fluoride ion is negatively charged and naturally likes to react with positively charged ions in the body like calcium and magnesium. When fluoride and calcium bind, creating a “precipitate,” a life-threatening condition of decreased calcium (hypocalcemia) can occur. Left untreated, decreases in calcium (and magnesium) can cause abnormal heart rhythm leading to heart attack, muscle spasms and death. Calcium administration is the main treatment for HF toxicity [28].
13. 1112°F - Octafluorocyclobutane (OFCB) [21]: Octaflurocyclobutane is a fluorine-containing gas that is used in the semiconductor industry, sold as Zyron 8020 by DuPont. According to DuPont, inhaling high levels of octafluorocyclobutane can cause heart beat irregularities, unconsciousness and death. People with pre-existing heart conditions may be extra vulnerable. Only a few toxicity studies in animals are available for octafluorocyclobutane. In one study, rats exposed to a one-time-only inhaled exposure of octafluorocyclobutane lost weight and had abnormal breathing. Dogs that inhaled high concentrations (10-25% air), and were dosed with the stimulant epinephrine, had heart problems. According to DuPont, tests for genetic damage in insects are “inconclusive” [29].
14. 1112°F - Perfluorobutane (PFB, Trade Name CEA-410) [21]: As a global warming chemical, perfluorobutane has a long half-life in the upper atmosphere and has over 8,000 times the global warming potential of carbon dioxide [30]. Perfluorobutane is not as acutely toxic as other PTFE off-gases, but has not been tested for long-term effects.
15. 1202°F - Carbon tetrafluoride (CF4, perfluoromethane) [21]: In addition to being a long-lived fluorinated Teflon “off-gas,” perfloromethane is used in the semiconductor industry, is a refrigerant and propellant and a byproduct of aluminum production. The U.S. government is encouraging these industries to decrease emissions of perfluoromethane because it is a potent greenhouse gas, with a global warming potential almost 6000 times higher than carbon dioxide, and can last in the environment for 50,000 years [30, 31]. In the past, perfluoromethane has been used in pesticides as an “inert” ingredient, [32] a label that has nothing to do with toxicity but only means the ingredient is not the main active pesticide.
Inhaling fluorinated hydrocarbons like carbon tetrafluoride can cause eye, ear and nose irritation; heart palpitations; irregular heart rate; headaches; confusion; lung irritation, tremors and occasionally coma [33].
References:
[1] Clayton, JW. 1967. Fluorocarbon toxicity and biological activity. Fluorine Chemistry Reviews 1(2): 197-252.
[2] Blandford, TB., Seamon, PJ., Hughes, R., Pattison, M and Wilderspin, MP. 1975. A case of polytetrafluoroethylene poisoning in cockatiels accompanied by polymer fume fever in the owner. Vet Rec 96(8): 175-8.
[3] Zanen, AL and Rietveld, AP. 1993. Inhalation trauma due to overheating in a microwave oven. Thorax 48(3): 300-2.
[4] Seidel, WC., Scherer, KV, Jr.., Cline, D, Jr.., Olson, AH., Bonesteel, JK., Church, DF., Nuggehalli, S and Pryor, WA. 1991. Chemical, physical, and toxicological characterization of fumes produced by heating tetrafluoroethene homopolymer and its copolymers with hexafluoropropene and perfluoro(propyl vinyl ether). Chem Res Toxicol 4(2): 229-36.
[5] Johnston CJ, Finkelstein JN, Mercer P, Corson N, Gelein R, Oberdorster G. 2000. Pulmonary effects induced by ultrafine PTFE particles. Toxicol Appl Pharmacol 168:208-15.
[6] Ellis DA, Mabury SA, Martin JW, Muir DC. 2001. Thermolysis of fluoropolymers as a potential source of halogenated organic acids in the environment. Nature 412:321-4.
[7] National Toxicology Program (NTP). 2002. 10th Report on Carcinogens. http://ehp.niehs.nih.gov/roc/toc10.html.
[8] Hazardous Substances Data Bank (HSDB). 2003. Full record for 1,1,2,3,3,3 - hexafluoro-1-propene (CASRN: 116-15-4). Available online at http://toxnet.nlm.nih.gov/.
[9] Environmental Protection Agency (EPA). Fluoroalkenes Test Results; Data submitted by DuPont on Hexafluoropropene (HFP). Office of Prevention, Pesticides & Toxic Substances Data Development (Testing) Policy Avialable online at http://www.epa.gov/oppt/chemtest/fluralke.htm.
[10] Environmental Protection Agency (EPA). List of other (inert) pesticide ingredients. Available online at http://www.epa.gov/opprd001/inerts/lists.html.
[11] Cornish J, Callon KE, Lin CQ, Xiao CL, Mulvey TB, Cooper GJ, Reid IR. 1999. Trifluoroacetate, a contaminant in purified proteins, inhibits proliferation of osteoblasts and chondrocytes. Am J Physiol 277:E779-83.
[12] Hunter ES, 3rd, Rogers EH, Schmid JE, Richard A. 1996. Comparative effects of haloacetic acids in whole embryo culture. Teratology 54:57-64.
[13] Buschmann J, Bartsch W, Dasenbrock C, Fuhst R, Pohlmann G, Preiss A, Berger-Preiss E. 2001. Cross-fostering inhalation toxicity study with HCFC-123 in lactating Sprague-Dawley rats. Inhal Toxicol 13:671-87.
[14] Cappon GD, Keller DA, Brock WJ, Slauter RW, Hurtt ME. 2002. Effects of HCFC-123 exposure to maternal and infant rhesus monkeys on hepatic biochemistry, lactational parameters and postnatal growth. Drug Chem Toxicol 25:481-96.
[15] Lantum HB, Baggs RB, Krenitsky DM, Anders MW. 2002. Nephrotoxicity of chlorofluoroacetic acid in rats. Toxicol Sci 70:261-8.
[16] Key BD, Howell RD, Criddle CS. 1997. Fluorinated organics in the biosphere. Environmental Science & Technology 31:2445-2454.
[17] Hazardous Substances Data Bank (HSDB). 2003. Full record for fluoroacetic acid (CASRN: 144-49-0). Available online at http://toxnet.nlm.nih.gov/.
[18] Integrated Risk Information System (IRIS). 1991. Sodium Fluoroacetate (CASRN: 62-74-8). Available online at http://www.epa.gov/iris/subst/0469.htm.
[19] Environmental Protection Agency (EPA). 2003. Preliminary risk assessment of the developmental toxicity associated with exposure to perfluorooctanoic acid and its salts. March 17, 2003.
[20] Environmental Protection Agency (EPA). 2002. Revised draft hazard assessment of perfluorooctanoic acid and its salts, November 4, 2002. U.S. EPA Administrative Record AR226-1136.
[21] Arito, H and Soda, R. 1977. Pyrolysis products of polytetrafluoroethylene and polyfluoroethylenepropylene with reference to inhalation toxicity. Ann Occup Hyg 20(3): 247-55.
[22] Hazardous Substances Data Bank (HSDB). 2003. Full record for silicon tetrafluoride (CASRN: 7783-61-1). Available online at http://toxnet.nlm.nih.gov/.
[23] Waritz, RS. 1975. An industrial approach to evaluation of pyrolysis and combustion hazards. Environ Health Perspect 11: 197-202.
[24] United States Department of State and Department of Commerce. 2003. U.S. Chemical Weapons Convention Web Site. Site sponsored by the United States Department of State (DOS), Bureau of Arms Control and the United States Department of Commerce, Bureau of Industry and Security (BIS) Available online at http://www.cwc.gov/.
[25] Patocka J, Bajgar J. 1998. Toxicology of perfluroisobutene. The Applied Science and Analysis (ASA) newsletter ISSN 1057-9419 Available online at http://www.asanltr.com/ASANews-98/pfib.html.
[26] Scheel, LD., Lane, WC and Coleman, WE. 1968. The toxicity of polytetrafluoroethylene pyrolysis products including carbonyl fluoride and a reaction product, silicon tetrafluoride. Am Ind Hyg Assoc J 29(1): 41-8.
[27] Hazardous Substances Data Bank (HSDB). 2003. Full record for carbon difluoride (CASRN: 353-50-4). Available online at http://toxnet.nlm.nih.gov/.
[28] International Programme on Chemical Safety (IPCS). 1995. Hydrogen Fluoride (CASRN: 7664-39-3). Available online at http://www.inchem.org/documents/pims/chemical/ hydfluor.htm#PartTitle:1.%20%20%20%20NAME.
[29] DuPont. 2002. Material Safety Data Sheet (MSDS) for Zyron (octafluorocyclobutane CASRN 115-25-3). Available online at msds.dupont.com/msds/pdfs/EN/PEN_09004a2f800a5e88.pdf.
[30] Environmental Protection Agency (EPA). Global Warming Potentials of ODS (Ozone Depleting Substances) Substitutes. Available online at http://www.epa.gov/ozone/geninfo/gwps.html.
[31] State Department. Mitigating climate change: methane and other greenhouse gas programs. Available online at http://www.state.gov/www/global/oes/97climate_report/part4b.html.
[32] Environmental Protection Agency (EPA). 1998. Inert ingredients no longer used in pesticide products. Federal Register (Volume 63, Number 121):Page 34384-34390 Available online at http://www.epa.gov/fedrgstr/EPA-PEST/1998/June/Day-24/p16571.htm.
[33] Hazardous Substances Data Bank (HSDB). 2003. Full record for tetrafluoromethane (CASRN: 75-73-0). Available online at http://toxnet.nlm.nih.gov/.
Heated pans get toxic in minutes
 EWG Experiment 1:
EWG Experiment 1:
Teflon® coated pan on a Whirlpool Gold Electric Range.
Test conducted on May 13, 2003 by Environmental Working Group. A 10" diameter aluminum Europa Basic Essential Everyday pan with a Non-stick Teflon® Classic interior was placed on the burner of a Whirlpool Gold electric range top. At time = 0 seconds, the pan temperature measured 73.5°F. The burner was turned up to the highest heat setting, and a timer was started. Using a Raytek Infrared Model Raynger ST Pro noncontact thermometer with an accuracy of +/- 2°F, the temperature was monitored every twenty seconds for 300 seconds (5 minutes). In this period of time, the temperature of the pan reached 721°F. At this point the pan was removed from the burner and the burner was turned off. The smell of burning plastic was noted.
EWG Experiment 2:
Non-stick coated pan on a Tappan Electric Range.
Test conducted on May 12, 2002 by a university food safety lab scientist. A 10" diameter aluminum skillet from Dollar General was placed on the burner of a Tappan electric range top. At time = 0 seconds, the pan temperature measured 72°F. The burner was turned up to the highest heat setting, and the timer was started. Using a Raytek Infrared Model Raynger ST noncontact thermometer with an accuracy of +/- 2°F, the temperature was monitored by scanning the thermometer back and forth within the pan to obtain an average temperature reading; this was done every twenty seconds for 200 seconds (3 minutes and 20 seconds). In this period of time, the temperature of the pan reached a maximum of 754°F, and an average pan temperature of 736°F. The temperature was still rapidly climbing. At this point the pan was removed from the burner and the burner was turned off. A small line of changed color in the interior of the pan was noted.
EWG Experiment 3:
Non-stick coated pan on a Kenmore Natural Gas Range.
Test conducted on May 12, 2002 by a university food Safety lab scientist. A 10" diameter aluminum skillet from Dollar General was placed on the burner of a Kenmore natural gas range top. At time = 0 seconds, the pan temperature measured 77°F. The burner was turned up to the highest heat setting, and a timer was started. Using a Raytek Infrared Model Raynger ST noncontact thermometer with an accuracy of +/- 2°F, the temperature was monitored every twenty seconds for 300 seconds (5 minutes). In this period of time, the temperature of the pan reached 664°F. At this point the pan was removed from the burner and the burner was turned off. A scent of “residual processing oils” was noted.

Teflon can't stand the heat
Download the infographic with references (PDF format)
Teflon offgas studies
Over the past five decades scientists from DuPont, government, and academia have published studies documenting temperatures at which non-stick cookware coatings begin to break apart, offgasing chemicals and particulate matter into the air. Dealing with multiple cases of polymer fume fever in their workers, DuPont scientists conducted a series of studies beginning in the 1950s to identify the toxic components from heated Teflon, killing birds and rats in efforts to understand the potency of the gases and particles.
Teflon Decomposition Products:
Studies show that thermal degradation of Teflon leads to the slow breakdown of the fluorinated polymer and the generation of a litany of toxic fumes including TFE (tetrafluoroethylene), HFP (hexafluoropropene), OFCB (octafluorocyclobutane), PFIB (perfluoroisobutane), carbonyl fluoride, CF4 (carbon tetrafluoride), TFA (trifluoroacetic acid), trifluoroacetic acid fluoride, perfluorobutane, SiF4 (silicon tetrafluoride), HF (hydrofluoric acid), and particulate matter. At least four of these gases are extremely toxic - PFIB, which is a chemical warfare agent 10 times more toxic than phosgene (COCl2, a chemical warfare agent used during World Wars I and II), carbonyl fluoride (COF2 which is the fluorine analog of phosgene), MFA (monofluoroacetic acid) which can kill people at low doses, and HF, a highly corrosive gas.
Many of the thermal degradation products are unmatched in their environmental persistence. Besides fire and heating, which are not considered normal methods of environmental degradation, some of these compounds have no known degradation methods, including four gaseous chemicals (TFA, PFOA, CF4, PFB) and some components of the particulate matter that are highly persistent. TFA and the other PFOA-like perfluorinated acids that have been detected in Teflon degradation studies have “no known significant loss mechanism” [1]. In addition, the perfluorinated alkene HFP, which makes up the bulk of the degradation products at temperatures above 680°F (360°C), will react with OH radicals in the troposphere to produce TFA with 100% conversion [2].
The lowest temperature at which nonstick coatings have been reported to kill birds in a peer-reviewed study is 396°F (202°C) [3]. In May 1998, poultry researchers at the University of Missouri recorded 52 percent mortality in 2400 chicks within three days of the birds being placed into floor pens with new PTFE-coated heat lamp bulbs. After ruling out bacterial infections like E. Coli and Salmonella, or toxic gases such as sulfur dioxide, carbon monoxide and carbon dioxide, the scientists finally linked the chick deaths to offgas products from the PTFE-coated bulbs. All of the chicks examined after death had lung lesions and moderate to severe pulmonary edema consistent with “PTFE toxicosis.”
The researchers also learned from a private communication with Dr. Bedros Nersessian that a duck research facility that used the same PTFE-coated heat lamps had 23.3 percent death in ducklings, over 400 ducklings in all, within five days [3]. In 1997, two English veterinarians reported a case in which eight raptor deaths over three months were attributed to PTFE coated heat lamps. This incident was described in a letter on PTFE toxicity in birds published in the journal Veterinary Record [4]. In addition, an inadvertent poisoning of pet birds has been reported when a Teflon-coated surface was heated to 325°F (163°C). | View Bird Death Diaries
These reports and personal accounts indicate that Teflon offgases toxic substances at temperatures as low as 396°F and 325°F. In 1991, a report by a collaborative team of DuPont and Louisiana State University scientists also addressed this issue. These scientists generated low molecular weight PTFE by heating Teflon to high temperatures and allowing the fumes to age for a few minutes; aging allowed the chemicals in the fumes to react to form small Teflon molecules. The scientists found that when this low molecular weight PTFE was reheated to 464°F two out of three exposed rats died. Evidence indicated that particulate matter was responsible for the rats’ death, and this particulate matter was composed of small molecules of Teflon [5].
Seidel and coworkers explain that “fumes generated at temperatures below 572°F (300°C) are formed exclusively by sublimation of a low MW (molecular weight) fraction already present in the polymer”[5]. From the Seidel study we can conclude that at low temperatures PTFE offgases particulate matter which is composed of small molecules of PTFE. We can also conclude that this particulate matter, which has been reported to cause bird deaths at temperatures below 500°F (260°C), is the result of the presence of low molecular weight PTFE.
This low molecular weight PTFE may be present in Teflon at the time of Teflon’s manufacture, or the PTFE may be created when a pan is heated repeatedly and the coating degrades as PTFE bonds break. PTFE particles have been measured in offgas products at temperatures as high as 1067°F (575°C).
According to Waritz's 1975 paper, particulate matter is emitted from Teflon pans between 554°F and 1067°F (290 and 575°C)[6]. These particles have been linked to bird deaths that can occur when an empty pan is heated on a burner. At these temperature the Teflon molecule breaks apart into smaller Teflon particles. When this occurs the carbon-carbon bonds of Teflon break, generating free radicals; these free radicals can then form alkenes, or they can react with oxygen to form carboxylic acids (forming PFOA, a chemical for which EPA is considering regulatory action, and PFOA-like compounds). The resulting particulate matter is thus a mixture of perfluorinated alkanes, perfluorinated alkenes, and perfluorinated acids [3, 7].
Many studies have established the toxicity of particulate matter generated at temperatures above 554°F (290°C). A study conducted by Zapp et al. in 1955 first indicated that particulate matter was implicated in the toxicity of PTFE [8]. In 1959, Clayton et al. found that PTFE resins will generate toxic products when heated in air. Clayton showed that the particulate matter is toxic when he demonstrated that the toxicity of the offgas products is removed when the PTFE offgas products are filtered to remove the particulate matter before animal exposure [9].
In 1968, DuPont scientists Waritz and Kwon showed that rats exposed to a 20 gram sample of PTFE heated to 842°F (450°C) produced death within four hours. The authors note that "exposures lasted four hours unless all rats exposed succumbed earlier," [10] but they failed to report the time to death among the rats. It was found that when the particulate matter was filtered from the offgas products before the rats were exposed, the mortality was reduced to zero; this provides evidence that the particulate matter is required for toxicity at 842°F. Based on the concentrations of particulate matter that caused rat death, Waritz and Kwon calculate that particulate matter is lethal at concentrations of only 1.4 parts per million (ppm) [10].
In a 1975 study by Waritz, Teflon breakdown products generated at lower temperatures were determined to cause death. Within four hours of exposure to a Teflon-coated pan heated to 536°F (280°C) parakeets died, and quail were killed when the pan was heated to 626°F or 330°C. These temperatures are easily accessible on a stovetop. Rats were killed within four hours when a Teflon pan was heated to between 797°F and 842°F (425 to 450°C). Once again, the DuPont scientist failed to report the time to death for the animals [6].
Many gases are also evolved when Teflon is heated. Heated to temperatures between 680 and 1112°F (360 to 600°C), Teflon generates TFE [1, 11]. This chemical is one of 174 chemicals that the National Toxicology Program considers to be reasonably anticipated human carcinogens [12]. Teflon heated to between 680 and 1202°F (360 and 650°C) will also generate HFP [1, 11]. Carbonyl fluoride, COF2, a toxic gas and the fluorinated cousin of the chemical warfare agent phosgene, is emitted from Teflon that is heated between 824°F and 1292°F (440 and 700°C) [6, 11]. PFIB, perfluoroisobutene, a chemical warfare agent that is ten times more toxic than phosgene, is detected when Teflon is heated in air between 887°F and 1004°F (475 and 540°C) [6]. Above 1202°F (650°C), carbon tetrafluoroide and carbon dioxide are the major products generated during the decomposition [7].
Other chemicals that have been detected in the offgas products of heated Teflon are trifluoroacetetic acid (TFA), difluoroacetic acid (DFA), and monofluoroacetic acid (MFA, also known as compound 1080, a pesticide and historical chemical warfare agent which has an LD50 of 0.7 – 2.1 mg/kg in men [13]), and various other perfluorinated acids with the general formula CF3(CF2)nCO2H; these acids were detected by University of Toronto scientists Ellis and Mabury (and coworkers) in a 2001 study in which Teflon was heated to 680°F (360°C) for 2 hours [1]. Finally, in the presence of glass (SiO2), SiF4 has been detected in the offgas products when teflon is heated to temperatures of 878°F (470°C)and higher [7, 11]; SiF4 is also a toxic gas and it has been reported that the presence of SiF4 enhances the toxicity of the other toxic Teflon offgas products [14].
In a recent PTFE rat toxicity study, Dr. Carl Johnston et al. found that neither the ultrafine particles or the gases produced by PTFE are toxic by themselves, but the two together are extremely toxic together [15]. The authors suggest the particles are acting as carriers for absorbed gas particles, allowing the gas particles to travel to the lower respiratory tract and cause severe damage. Alternatively, the particles could be generating toxic reactive groups on their surfaces. Also, the size of the particles matters. The most toxic particles are called ultrafine and are 16 nanometers in diameter. If the particles are able to coagulate and form larger particles, greater than 100 nm, they are not as acutely toxic to rats.
In one case of human polymer fume fever in the literature, the author reports a case in which a person developed polymer fume fever about one hour after a non-stick pan overheated. Five cockatiels in the house died within 30 minutes [16]. In another case, a healthy 26-year-old woman went to the hospital complaining of difficult breathing, chest tightness and cough after being exposed to toxic fumes coming from a defective microwave oven part: a melted and scorched Teflon block used as an axle for a rotating platform in the oven. At the hospital, doctors noted that her heart was racing, and she had high blood pressure, increased white blood cell count (leukocytosis) and was breathing heavily. An X-ray showed she had “diffuse pulmonary infiltrate.” Her lung function was still abnormal a month later. This woman's two pet parakeets died within minutes of being exposed to the Teflon fumes [17].
References:
[1] Ellis, DA., Mabury, SA., Martin, JW and Muir, DC. 2001. Thermolysis of fluoropolymers as a potential source of halogenated organic acids in the environment. Nature 412(6844): 321-4.
[2] Machino, M. 2000. Atmospheric chemistry of CF3CF=CF2: kinetics andmechanism of its reaction with OH radicals, chlorine atoms, and ozone. J. Phys. Chem. A 104: 7255-7260 (As cited in: Ellis, DA., Mabury, SA., Martin, JW and Muir, DC. 2001. Thermolysis of fluoropolymers as a potential source of halogenated organic acids in the environment. Nature 412(6844): 321-4).
[3] Boucher, M., Ehmler, TJ and Bermudez, AJ. 2000. Polytetrafluoroethylene gas intoxication in broiler chickens. Avian Dis 44(2): 449-53.
[4] Forbes, NA and Jones, D. 1997. PTFE toxicity in birds. Vet Rec 140(19): 512.
[5] Seidel, WC., Scherer, KV, Jr.., Cline, D, Jr.., Olson, AH., Bonesteel, JK., Church, DF., Nuggehalli, S and Pryor, WA. 1991. Chemical, physical, and toxicological characterization of fumes produced by heating tetrafluoroethene homopolymer and its copolymers with hexafluoropropene and perfluoro(propyl vinyl ether). Chem Res Toxicol 4(2): 229-36.
[6] Waritz, RS. 1975. An industrial approach to evaluation of pyrolysis and combustion hazards. Environ Health Perspect 11: 197-202.
[7] Coleman, WE., Scheel, LD and Gorski, CH. 1968. The particles resulting from polytetrafluoroethylene (PTFE) pyrolysis in air. Am Ind Hyg Assoc J 29(1): 54-60.
[8] Zapp, JA., Limperos, G and Brinker, KC. 1955. Toxicity of Pyrolysis Products of Teflon Tetrafluoroethylene Resin. Amer. Indust. Hyg. Assoc., Annual Meeting, May 1955. (As cited in: Waritz, RS and Kwon, BK. 1968. The inhalation toxicity of pyrolysis products of polytetrafluoroethylene heated below 500 degrees centigrade. Am Ind Hyg Assoc J 29(1): 19-26).
[9] Clayton, JW., Hood, DB and Raynes-Ford, GE. 1959. The Toxicty of the Pyrlysis Products of Teflon TFE-Fluorocoarbon Resins. Amer. Indust. Hyg. Assoc. Annual Meeting, May 1959. (As cited in: Waritz, RS and Kwon, BK. 1968. The inhalation toxicity of pyrolysis products of polytetrafluoroethylene heated below 500 degrees centigrade. Am Ind Hyg Assoc J 29(1): 19-26.
[10] Waritz, RS and Kwon, BK. 1968. The inhalation toxicity of pyrolysis products of polytetrafluoroethylene heated below 500 degrees centigrade. Am Ind Hyg Assoc J 29(1): 19-26.
[11] Arito, H and Soda, R. 1977. Pyrolysis products of polytetrafluoroethylene and polyfluoroethylenepropylene with reference to inhalation toxicity. Ann Occup Hyg 20(3): 247-55.
[12] National Toxicology Program (NTP). 2002. 10th Report on Carcinogens. http://ehp.niehs.nih.gov/roc/toc10.html.
[13] Key, BD., Howell, RD and Criddle, CS. 1997. Fluorinated organics in the biosphere. Environmental Science & Technology 31(9): 2445-2454.
[14] Scheel, LD., Lane, WC and Coleman, WE. 1968. The toxicity of polytetrafluoroethylene pyrolysis products including carbonyl fluoride and a reaction product, silicon tetrafluoride. Am Ind Hyg Assoc J 29(1): 41-8.
[15] Johnston, CJ., Finkelstein, JN., Mercer, P., Corson, N., Gelein, R and Oberdorster, G. 2000. Pulmonary effects induced by ultrafine PTFE particles. Toxicol Appl Pharmacol 168(3): 208-15.
[16] Blandford, TB., Seamon, PJ., Hughes, R., Pattison, M and Wilderspin, MP. 1975. A case of polytetrafluoroethylene poisoning in cockatiels accompanied by polymer fume fever in the owner. Vet Rec 96(8): 175-8.
[17] Zanen, AL and Rietveld, AP. 1993. Inhalation trauma due to overheating in a microwave oven. Thorax 48(3): 300-2.
DuPont spin
DuPont has repeatedly misled consumers about the safety of their products, publishing misleading information, obscuring important safety warnings, and intimidating bird-related websites which provide pet owners with warnings about the dangers Teflon poses to pet birds.
References
[1] DuPont 2001. University of Toronto Study and Cookware Safety. Accessed online at http://www1.dupont.com/NASApp/dupontglobal/corp/index.jsp? page=/content/US/en_US/news/position/cookware_safety.html. May 11 2003.
[2] DuPont 2003. "Consumer products help: Cookware safety. Will cooking fumes generated while cooking with non-stick cookware harm people or animals, especially pet birds?" Accessed online May 10 2003 from http:/www.teflon.com.
[3] Boucher M, Ehmler TJ, Bermudez AJ. 2000. Polytetrafluoroethylene gas intoxication in broiler chickens. Avian Dis 44:449-53.
[4] Stewart Bob. 2002. Personal communication with Dr. Jennifer Klein, Environmental Working Group. May 9, 2002.
[5] Stewart Bob. 2002. Personal email communication with Anne Morgan, Environmental Working Group. [date]
[6] Waritz, R.S. 1975. An industrial approach to evaluation of pyrolysis and combustion hazards. Environ Health Perspect 11:197-202.
[7] DuPont 2003. "Making a safe home for your bird," written by Peter S. Sakas. Accessed online May 8 2003 from http://www.dupont.com/teflon/newsroom/bird.html.
- In a recent news release, DuPont claimed that its coating remains intact indefinitely at 500°F: "DuPont non-stick coatings will not begin to deteriorate until the temperature of the cookware reaches about 500 degrees F, (260 degrees C), and significant decomposition of the coating will occur only when temperatures exceed about 660 degrees F (340 degrees C). These temperatures alone are well above the normal cooking range" [1][2]. Experiences of consumers whose birds have died from fumes generated at lower temperatures show that this is not the case. In one case researchers at the University of Missouri documented the death of about 1,000 broiler chicks exposed to offgas products from coated heat lamps at 396°F [3]. In another case a bird died after its owner preheated a new Teflon-lined oven to 325°F [4][5].
- DuPont also claims that human illness will be produced only in cases involving gross overheating: "In cases where non-stick coating is grossly overheated (any food would have long been burned to an inedible state at this point), fumes may produce temporary flu-like symptoms." [2]. Yet DuPont's own scientists have concluded that polymer fume fever in humans is possible at 662°F, a temperature easily exceeded when a pan is preheated on a burner or placed beneath a broiler, or in a self-cleaning oven [6].
- Although DuPont has known of the particular susceptibility of birds to Teflon offgas products for decades, and the hazards Teflon poses to pet birds in the home, the company recently employed intimidation tactics to stop bird-related websites from discussing the toxicity of Teflon fumes to birds, impressing one webmaster to the point that he changed all mentions of "Teflon" in his public service messages on bird safety to "a product whose name I can no longer use due to its trademark."
>>| Read a letter sent to a bird owner from DuPont - DuPont publicly acknowledges that Teflon can kill birds, but the company-produced public service brochure on bird safety discusses the hazards of ceiling fans, mirrors, toilets, and cats before mentioning the dangers of "PTFE" fumes at the bottom of the two-page brochure ("Teflon" is not mentioned) [7].
Bird death diaries
Teflon and other non-stick coatings can produce fumes that kill birds. Below we recount a small fraction of the documented cases of bird deaths attributed to fumes and particles released from non-stick coatings on cookware, heat lamps, and other home appliances.
• Documented pet bird deaths in peer-reviewed literature
• Mass wild bird deaths from non-stick manufacturing fumes
• From the vet's mouth: bird death cases
• 100 websites that discuss birds and Teflon toxicosis


Killed: Watson, Dee-Dee, and more than 50 other birds
Implicated in death: Overheated, electric non-stick pan
In December 2002 a family lost more than 55 birds after a pan on their stove overheated when the water burned off. Despite their efforts to ventilate the house after they noticed the smoldering pan, one by one they lost three Blue and Gold Macaws, a Camelot Macaw, one Scarlet Macaw, 2 sun conures, 2 nanday conures, 2 TAGS, 4 parrolets, and over 40 cockatiels. [1]
Theresa K. writes:
I awoke to my Husband calling me to get up, something was wrong with Watson. (Watson, was a rescued Blue and Gold, that had become my life.) I jumped out of bed and in a macabre move my husband turned around, craddling Watson and said, Watson is dead. I thought my world had stopped right there.
I carried Watson into the bathroom, and rocked and cried for at least 20 minutes. I gained some composure and said, I have to put him in the freezer... we have a full breeding aviary, and I knew the responsible thing to do was to do a necropsy.
As I walked into the kitchen, I saw the pan that was smoldering. The pan had been full of water and the burner turned off, but a short circuit had turned it on even though the handle/ indicator said it was off. I then noticed a sun conure flying by and dropping to the ground, also, immediately dead.
The next few hours are the hardest I will probably ever go through. One by one the birds were dying. Agonizing screaming... we opened up everything in the house. It was snowing and freezing. I placed fans everywhere to exhaust the fumes. (I lost 2 Boston Terrier puppies from pneumonia 2 days later)... We did our best to hold each and every one of the macaws as they were dying, I didn't want them to be alone in their pain. In my mind will always be etched my husband holding the Caleb, the Camelot Macaw, and Max, a Nanday tucked under his shirt, as I went down the steps to pull DeeDee, a 50+ Blue and Gold and hold her as she was screeching her last breaths away.... My husband sounded so vulnerable as he screamed out, "Oh God, Not DeeDee."
Thankfully, most is still a blur. That night we lost 3 Blue and Gold Macaws, The Camelot Macaw, one Scarlet Macaw, 2 sun conures, 2 nanday conures, 2 TAGS, 4 parrolets, and over 40 cockatiels. The next morning we had to tell the kids and they did not deal well with this. My 6 year old put a box over his head, that was there almost constantly for a week, saying he was a super hero... DR Thunder...and he would grill me, why did I kill the birds? Why did I buy the pan, why couldn't I save them?
My 8 year old daughter became withdrawn and drew a grave stone marker on her wall with the date and then in small print, all of the names of the birds lost... It is still there, I can't bring myself to cover it up. I saw their pain for months in their school work and their pretend playing. Well meaning friends have been dropping by and bringing me "free" misfit birds....
I have had to ask them to stop, we are running out of room. So, I have a houseful of screaming noisy birds again, but they can't drown out the noticable void of certain voices. I am still numb. It put a great strain on our marriage and I am depressed a lot.... not even zoloft is lifting my or my husband's dark cloud of pain.Please if there is anything I can do to help, please call on me, I will gladly give any time you need. Thank you for listening, I didn't realize how many tears were still in me.
Killed: Moluccan cockatoo
Implicated in death: Teflon-coated iron and carpet glue
A woman reported the death of a moluccan cockatoo four hours after contractors repaired a carpet tear using carpet glue and a Teflon iron [2]. Michelle writes:
In April we had a small piece of carpet cut out and replaced in the living room. I notified the repair man that I have birds in my house and cannot have any fumes, teflon used in my home. I have an amazon and a moluccan cockatoo. They assured me that there were no fumes or toxic materials being used. Four hours later my moluccan Roxy started to throw up, we rushed her to an emergency 24 hr. vet who didn't know a thing about birds. She gave her a injection of antibiotic and she soon after went into respiratory distress and died at 1:15 am. We did not do a necropsy on her. I found out the next day they had used carpet glue and a Teflon iron to repair the carpet. Even though I took Roxy upstairs to another room while the work was being done she died anyway. A part of us died too that night. I'll never know for sure exactly what she died from, but I believe this was the cause. Many people have asked my then why is my amazon OK? I don't really know. Let everyone know you cannot trust workers in your house, they don't know what they are using in your house and they know less about parrots!!!
Excerpted from http://www.birdhotline.com/comment.htm
Killed: Ringneck, two Amazons, and a blue and gold macaw
Implicated in death: Skillet and electric space heater
An Iowa family lost a ringneck, two Amazons, and a blue and gold macaw within ten minutes of using an electric skillet and a space heater concurrently while preparing dinner. As the bird deaths progressed, an avian veterinarian questioning the bird owner over the telephone told her to turn off the space heater, which was likely coated with PTFE. By necropsy a local veterinarian confirmed lung damage in the dead birds consistent with that caused by inhalation of PTFE fumes. [3]
I received a truly heartbreaking call a couple of weeks ago from a very nice lady and a true bird lover named Dawn Costello. Dawn lives near New Virginia, Iowa...her birds died even though she didn't make any mistakes.
Most of us know by now that (a product name I can no longer use due to it's trademark), when heated over approximately 500 degrees will emit fumes that will kill birds. Dawn knew this too, and, like me, she used an electric (a product name I can no longer use due to it's trademark) skillet with a heat control and never set the control over 300 degrees. There have been some reports of (a product name I can no longer use due to it's trademark) related bird deaths in which the temperature of the cooking surface was not overheated but the surface was old and scarred and apparently released fumes through the marred area. This did not apply in Dawn's case since the cooking surface of her skillet was in good shape.
Dawn's family was preparing for dinner one evening using the electric skillet. An electric space heater was also running in the area since it was a chilly evening. (Store that; it's important). She then noticed that her Ringneck was not visible in his cage. She had seen him playing there just 10 minutes before but she checked anyway just to make sure he was still in the cage and hadn't gotten out. She was horrified to find the bird lying dead on the floor of the cage. A moment later she heard a scream from a daughter in another room and rushed there to find her two Amazons dead. She then ran to her Blue and Gold Macaw's cage to find that bird nearly dead. (Tears are streaming down my face as I write this because, like many of you, I can put myself in Dawn's position. She lost half a dozen beloved members of her family in the space of 10 minutes.) She shut off the (a product name I can no longer use due to it's trademark) skillet (but not the electric heater) and made several frantic calls for help. Local vets were not available (why does that not surprise me?), but she was soon in contact with a national animal poison control center. An out-of-state avian vet returned the call about the same time the Blue and Gold went into convulsions. The vet immediately asked about (a product name I can no longer use due to it's trademark) and Dawn told him they had been using an electric skillet, but it was in good condition and on low heat. He then asked if there was an electric space heater running in the area. When told there was, he said, "Shut it off quick!"
ACCORDING TO THIS VET, (a product name I can no longer use due to it's trademark) IS BEING USED IN MORE AND MORE APPLIANCES FOR THE HOME. IN ADDITION TO SKILLETS AND ELECTRIC HEATERS, IT IS ALSO FOUND IN IRONS, HAIR DRYERS AND APPROXIMATELY 20 OTHER DEVICES.
The vet told Dawn that probably a small amount of fumes was leaking from the skillet and coupled with what was spilling from the heater, it was sufficient to kill her birds. The Blue and Gold succumbed within a few minutes. Dawn has had the (a product name I can no longer use due to it's trademark) diagnosis confirmed by Dr. Riordan who did necropsies on all the birds. He told her that even if she had been across the street from a vet, nothing could have saved her Macaw. Apparently the fumes cause hemorrhaging in the lungs and air sacs and once the symptoms are evident, it is usually too late.
Excerpted from http://www.quakerville.com/qic/ezine/96Issue5/qteflon.htm
Respiratory distress: Macaw
Implicated: Toaster oven
Karen Grahame witnessed the toxicity of Teflon fumes to her Macaw when she used her small oven to cook her dinner on two separate occasions. On the first occasion her Macaw experienced severe respiratory distress two hours after Karen broiled a steak in her oven. She rushed her ailing bird to the veterinarian who saved his life and told her that her pet would have died had she not brought him in when she did. At this point neither Karen nor the doctor suspected the oven. The macaw did not show signs of respiratory stress again until she used the same oven at 350 degrees. Karen Grahame now keeps her oven in the garage. She notes that the oven user manual does not indicate that the oven is coated with PTFE [6].
For energy reasons, I do not routinely use my gas oven to cook small items; I use a small "Toaster/broiler" oven whose interior does not have the PTFE appearance. The manual that came with it does not indicate PTFE components, although I have heard (since the experience I am about to relate) that heating elements are usually coated with PTFE. I have used this small oven for nearly two years without problem. Last summer I broiled a small steak in it the exact same way I had done many times previously; after finishing my dinner I brought my 4 birds out for their evening playtime with me, which lasts two hours. As I put them back in their cages, I noticed the macaw was in some kind of distress, breathing quite rapidly with his mouth open, which I had never experienced with a bird. None of the other birds were affected. I was able to get the macaw to an avian vet within 75 minutes of noticing the respiratory distress, and the vet treated him with oxygen and steroids. The macaw recovered with no apparent after-effects, but neither the vet nor I were able to pinpoint the cause, and the vet told me that the macaw would have died if I had not gotten him to the vet as quickly as I did. We initially dismissed the oven because none of the other birds were affected. However, there was a second similar occurrence of respiratory distress with the macaw, again immediately after using the oven (although this time it was not at broiling temperature, it was used at 350 degrees). Again, none of the other birds were affected, and again the macaw recovered. (I now use the oven outside in the garage!)
The vet has diagnosed the bird with "Respiratory Hypersensitivity Syndrome", basically the avian form of asthma/allergies, but even if this is so I still think that the oven gave off some kind of toxic fumes to trigger the attacks, as the oven was involved in both events. On the other hand, if it was PTFE, it had to have been a VERY small amount because none of the other birds were affected, and from what I know of PTFE it is fatal to birds in even very small amounts. Another possibility is a toxic fume that was not PTFE but still dangerous enough to affect a bird with an already-compromised respiratory system. (And if anything is deadly enough to kill a bird, it can't be good for people either, can it? Interestingly enough, I also have a compromised respiratory system, and I often wonder if there is a connection there as well.)
In any event, I have since read that ANY item that produces heat--hair dryers, coffeemaker heating plates, space heaters--potentially have PTFE treated components, and that the manuals do not necessarily indicate that is the case. It would be wonderful if you could convince manufacturers to post warning labels on the items or at least in the manuals if there is PTFE anywhere. It would be even better if research could find an alternative!
Good luck to you in your research,
Karen Grahame
Killed: Baby parrots
Implicated in death: New non-stick-lined Amana oven
After updating their kitchen with all new appliances, Bob and Lynnette Stewart prepared biscuits in their kitchen while their newborn baby parrots slept in their brooders on the kitchen counter. As they preheated the oven all of their parrots died. The Stewarts have brought a lawsuit against Amana, the manufacturer of their oven. As an exotic bird buisiness owner, Bob Stewart knew of the dangers of Teflon, and he has never brought an item into his house that he knew was Teflon coated, but, he said, “there was no warning on the paperwork (that their oven had Teflon coating).” (Temperature and time of death was learned in a private communication with Bob Stewart.) [7,8]
My name is Bob Stewart. My wife and I own a exotic bird business in Riverside, California called SOS4BIRDZ. Along with all of the bird products we sell and distribute we raise several species of parrots. We had an incident which we are still in litigation over. What happened was that our house was severely damaged in a fire and had to be gutted and re-built. In the process of the reconstruction we decided to upgrade all of the major built-in appliances. When we moved back in the house after the re-construction was completed we had several babies that had hatched and we were hand feeding them. All of our baby parrots were being kept in brooders on the kitchen counter. In the process of having them there we decided to use our new stove to bake something in the oven. Unknowingly we had no idea that the oven was in fact coated with a non-stick chemical. There was no warning on the paperwork concerning this. We were aware of such products as Teflon coated pots and pans being detrimental and deadly to birds and have made it a point to never have any of these type of products in the house. In the process of baking the food the toxins created by the heating of the oven were released and all of the babies died as a result of it. We have had an attorney going after Amana for the past year about this incident and we have yet to resolve it. Hopefully we can be of assistance in this matter since we have first hand experience with this terrible scourge.
Sincerely,
Bob Stewart,
SOS4BIRDZ, Inc.
Killed: Kola Bird, a Yellow Cheek Amazon Parrot
Implicated in death: Pan from which water had boiled off
A bird owner reported that her Yellow Cheek Amazon Parrot was killed as she heated some water for a cup of hot cocoa. The water boiled off and her bird died. [9]
I just lost my Kola Bird (he was a Yellow Cheek Amazon) to Teflon poisoning. I came home from work and was heating up some water for a cup of hot cocoa. I forgot about the water on the stove. A while later I heard my boy fall off of his perch, but I didn't hear anything else so I went to investigate. He was sitting on the bottom of the cage with his head kind of bobbing back and forth. In less than five minutes, he was dying in my arms. It was the saddest thing watching my friend of twenty-one years take his last breath in my arms. Please pass this piece on so that maybe another death can be prevented. It hurts real bad.
Killed: Pumpkin (a Moluccan cockatoo), a peach-faced Lovebird, and another Moluccan
Implicated in death: Nonstick grill plate on gas stove
Another bird-loving couple lost three pets in 1992 and 1993. Each bird died after steaks or “a large amount of food” were grilled on a Teflon-coated grill plate built into her gas cook-top stove. A Moluccan died after the steaks had been cooked while he sat in a room adjoining the kitchen. They found their peach-faced Lovebird dead on the floor above the kitchen two hours after grilling was complete. The third bird, their couple’s replacement Moluccan, died in the room adjoining the kitchen while the food was still being cooked. Necropsy indicated that this bird had been poisoned from exposure to Teflon fumes. The owners of these pets had specifically selected the stove because they were told that it did not have Teflon or similar non-stick surfaces. After the first bird death, they had suspected Teflon poisoning and again inquired whether their stove had any non-stick coating. They were again told that it did not. It was only after a thorough investigation of the manufacturer’s book that the owners learned that the built in grill plate was coated with a non-stick surface. [10]
Over the past year and a half, we've lost three pet birds from the use of a Teflon-coated grill plate on the gas cook-top stove in our house. The first incident occurred in May 1992. We lost an 8-year-old Moluccan cockatoo, Pumpkin, while I was cooking. The Moluccan was atop a large exercise gym in the family room, which adjoins the kitchen. A large amount of steaks were grilled on the gas grill portion of the cook top stove. At about 11:30, the Moluccan lost his balance and fell off the exercise gym. He fell about 2 feet. I rushed to him to check for injuries. He was having difficulty holding his balance and maintaining a grip. Approximately 10 minutes later, the bird died. We pulled into the parking lot of Actin Animal Hospital on Portsmouth Blvd as he took his last breath. They performed an autopsy, but the results were inconclusive because we believe the autopsy was performed incorrectly.
The second incident occurred in May 1993, while we were again cooking a large amount of food on the grill of the stove. This time we lost a male peach-faced lovebird. The conditions were nearly the same. I went upstairs about two hours after our guests left and discovered the lovebird dead in his cage. His mate was alright and showed no signs of stress other than the loss of her mate. No autopsy was made.
The third incident happened in mid-July. Again, a large amount of food was being grilled on this stove. A Moluccan that had replaced the bird we lost was on his perch 6' off the floor in his large cage in the foyer just off the kitchen. Our son passed by the cage and called to me that the bird was having difficulty breathing. I took him out of the cage, his grip being weak, and put him in a room that had been closed. We moved an air nebulizer already in operation for an Umbrella cockatoo that had aspergillosis into the room and tried to administer air to him. The bird was dead within 15 minutes. We took this bird to Dr. Ruth Ann MacQueen for an autopsy. She performed an observation exam and sent off specimens for detailed tests. Lab tests confirmed her findings: toxic poisoning consistent with exposure to heated Teflon.
We had originally selected this stove for installation in our new house in early 1992 because it did not have any kind of non-stick cooking surfaces. When we first inquired of the store saleslady, she said that the stove did not have any Teflon or similar non-stick coating surfaces. We suspected that Teflon was the culprit when the first bird died, at which time I had gone back to the saleslady and explained all about non-stick surfaces and birds in great detail. I wanted to reconfirm that there was no Teflon anywhere on the stove. Once again, we were told no. A few months later, I wanted to clean the oven and wanted the store to check with the manufacturer about Teflon being on any of the stove parts, even on its screws or washers, or the underside. The cleaning section of the manufacturer's book, which is the last 2-3 pages at the end of the book, mentioned that the cooking grills were covered with a non-stick surface. The store our builder got the stove from was A&B Propane on Military Highway.
Killed: Two canaries, two budgies
Implicated in death: Poaching pan
In 2001 a New Zealand family lost two canaries and two budgies after poaching eggs on a non-stick pan. These four brought to seven the number of birds that had died in the house over a period of seven years. In this instance, the bird owner requested an autopsy, which showed lung hemorrhages and congestion, in a pattern consistent with Teflon toxicosis. The deaths were attributed to a set of Teflon-coated pans purchased around the time the bird deaths began. The Ministry of Health took the case on for investigation [11].
Bird Deaths Linked to other Perfluorochemicals
Anecdotal reports indicate that other perflurochemical products are toxic to birds. One such incident is related below, involving a sofa treated with Stainsafe, a stain and water-repelling product similar to Dupont's Stainmaster. 3M Corporation reportedly received numerous reports, going back decades, of bird deaths caused by expossure to the original formulation of its Scotchgard stain and water repellent. The active ingredient in that product, PFOS, was forced from the market by EPA in 2000 (See EWG's report on PFCs on our main Web site and the Scotchgard story in our Chemical Industry Archives.)
Killed: Squigmund, a Mexican Redhead Amazon Parrot
Implicated in death: Stainsafe coating on new couch
Diane Sehnal, owner of a Mexican Redhead Amazon Parrot, purchased a new couch from Hansen’s Furniture Store in Winton, California. When she learned that her couch was treated with Stainsafe fabric protector, she inquired about its safety to birds and was told that it would be harmless to her pet of 16 years. Her parrot died 36 hours after her new couch was delivered. A necropsy revealed lung lesions, and an enlarged heart and spleen, pointing to the possibility of inhalation toxicity from Stainsafe. The typical lifespan of Amazon Parrots is 80 years. [4,5]
This is my first time on this site. I want to share an experience I recently had that caused my beautiful Mexican Red Head to die.
I purchased a new sofa from Hansen's Furniture Store in Winton, California. The sofa was treated with a fabric protector called STAINSAFE. I was told that stainsafe was harmless to birds. Thirty-six hours after the sofa was delivered my beloved SQUIGMUND died. On the morning of the second day that I owned the sofa Squigmund began to have labored breathing, and he was weak. I rushed him to his Avian vet. Squigmund had absolutely no strength to stand up in his carrying case so I had to clutch him to my chest while driving a five speed car. I was crying. I knew Squigmund was dying so I tried to collect myself, and tell him how much I loved him and I thanked him for all the joy, and comfort he gave me. He died 4 hours later at the vet's office.
I saved my money for a new sofa and I wanted my home to be cozy for myself, and Squigmund. Look at what I have done. I allowed this deadly sofa into my home which killed my darling sweet boy Squigmund. My companion for 16 years. He could sing "I left My Heart in San Francisco." He loved to flirt with all the girls young and old alike. He would whistle at them! He could laugh and he loved having his head scratched. He was a true comedian.A necropsy was perfomed by his usual vet who reported that Squigmund had leisions all over his lungs with an enlarged heart, and spleen. She said that he was poisioned to death. We decided to have a pathologist in sacramento who specializes in these areas of causes of death to birds. She confirmed that the cause of death was INHALATION TOXICITY due to the new sofa that was treated with Stainsafe.
The pathologist said that not only is Stainsafe a killer to birds but also related that new furniture can sometimes emit gases that are deadly to birds as well.
Squigmund was my only Parrot, and you can imagine the quality of our relationship. I cry every single day since he died on April 4, 2002. For as long as I shall live, I will never again experience loving a parrot like I did with Squigmund.
I have hired an animals rights lawyer in San Francisco. She was appalled at what happened to Squigmund. We will be demanding that warning labels be placed on this product Stainsafe to warn other companion bird owners of the dangers.
Thank you for reading this. Please tell others about this toxic killer Stainsafe... I do not want any other bird to ever suffer a heinous death like my Squigmund did.
Peer-Reviewed Bird Deaths


Killed: Five cocatiels
Implicated in death: Frying pan, accidentally overheated
In 1975, in one of the early peer-reviewed articles on bird deaths, the authors describe the deaths of five pet birds following the owner’s heating a non-stick (PTFE-coated) pan:
“Five cocatiels (Nymphicus hollandicus) died within 30 minutes following exposure to fumes from a frying pan coated with the "non-stick" plastic polytetrafluoroethylene (PTFE) that had accidentally overheated. Within an hour the owner developed symptoms of "polymer fume fever" but recovered in the next 24 hours. Clinical signs and post mortem lesions of the cockatiels are described and reference is made to the unusual susceptibility of parakeets to the pyrolysis products of frying pans coated with PTFE.” [12].
Killed: More than 1200 broiler chicks
Implicated in death: PTFE-coated heat lamp lightbulbs
Bird deaths related to non-stick coatings are not restricted to exotic species in the home. A recent article recounts that hours after moving 2400 broiler chicks to a research warehouse at University of Columbia-Missouri, researchers noticed that substantial numbers of chicks were dying. Four percent of the chicks died in the first four hours, and within 72 hours more than half of the chicks were dead. After investigating the possibility of many common gas toxicants, scientists traced the deaths to lightbulbs coated with the Teflon chemical PTFE: “Further investigation revealed that the only change in management practice in this facility prior to the onset of the severe mortality problem was the replacement of 48 heat lamp bulbs (one for each pen). The new heat lamp bulbs were polytetrafluoroethylene (PTFE) coated. PTFE gas intoxication has been reported in several exotic avian species, but this intoxication has not been previously reported in a poultry flock.” [13].
Killed: Two parakeets
Implicated in death: Melted Teflon microwave component
A Dutch doctor reported on the deaths of two pet parakeets that died within minutes of being exposed to toxic fumes coming from a defective microwave oven part — a melted and scorched Teflon block used as an axle for a rotating platform in the oven. The owner, a 26-year-old, healthy woman, went to the hospital complaining of difficult breathing, chest tightness and cough. At the hospital, doctors noted that her heart was racing, and she had high blood pressure, “leukocytosis” and was breathing heavily. Also, an X-ray showed she had “diffuse pulmonary infiltrate”. Her lung function was still abnormal a month later [14].
Killed: 80 to 100 wild birds
Implicated in death: Fumes from non-stick manufacturing facility
In 1997 scientists in England reported that 80 to 100 wild birds were found dead in a four-hour time span approximately 700 feet from an industrial plant in western England that coated sheet metal with non-stick paint containing PTFE. On the afternoon of the bird deaths, the plant had some problems that resulted in oven temperatures raised up to 880 degF (470 degC) to improve the coating of the sheet metal with the PTFE-containing paint. Wind direction was consistent with fumes travelling from the plant to where the dead birds were found. The authors write, "Most of the birds were found dead, but some were alive, gasping for breath, and died shortly afterwards" [15].
From the vet's mouth: bird death cases


Killed: 14 pet birds
Implicated in death: Teflon-coated stovetop burner drip pans
A Chicago-area woman lost 14 birds within 15 minutes of preheating four burners on her electric stove while preparing Thanksgiving dinner. A local avian veterinarian found lung damage (necrotizing pneumonitis) in a post mortem investigation, and upon questioning the bird owner in detail, attributed the deaths to fumes from new, Teflon-coated drip pans installed two weeks earlier [7][8]. Neither the Food and Drug Administration nor the Consumer Product Safety Commission has banned the use of PTFE coatings on drip pans, which DuPont studies show can approach 1000 degF during routine use. [17,18]
Killed: 107 chicks
Implicated in death: Teflon cookie sheet under over broiler
Chicago-area bird specialist Dr. Peter Sakas documented a case in which 107 chicks died after their owner put a Teflon cookie sheet under a broiler to catch drippings. [18].
Killed: All birds in a breeding operation
Implicated in death: Overheated Teflon pan
Dr. Robert Eckroade, director of the poultry pathology laboratory at New Bolton Center of the University of Pennsylvania School of Veterinary Medicine, reported a case he oversaw in which all the birds in two rooms of a breeding operation died after a Teflon pan in a home was overheated [17].
Killed: $2,000 pet bird
Implicated in death: Self-cleaning oven
Bird specialist Dr. Richard Nye of the Midwest Bird and Exotic Animal Hospital in west suburban Westchester oversaw a case in which a $2,000 bird died when the bird owner used the self-cleaning feature on a home oven.[17].
Killed: two pet birds
Implicated in death: non-stick pizza pan
Dr. Peter Sakas, a Chicago area veterinarian and bird expert, recounted a case in which an area woman lost a cockatiel and one other bird (species unnamed) while she heated pizza on a non-stick pan [17].
Killed: 14 pet birds
Implicated in death: Teflon cookie sheet used to bake bread
Dr. Sam Vaughn, an avian veterinarian specialist in Louisville, Kentucky, investigated a case in which 14 birds died within 30 minutes of their owner cooking bread on a Teflon cookie sheet and barely burning the bottom of the bread. The cookie sheets had been used before without incident. Dr. Vaughn’s autopsy of two of the 14 birds confirmed lung lesions that are a hallmark effect of PTFE toxicosis [19].
Wild Bird Deaths


Killed: Wild birds
Implicated in death:Fumes from cookware coating facility
In August 1996 the Canadian Cooperative Wildlife Health Centre (CCWHC) in Ontario, a collaboration of four university veterinarian colleges, investigated a report from the City of Scarborough Department of Public Health alleging an unusual number of wild bird deaths in an area bordering a cookware coating facility. Between July and October the CCWHC collected dead house sparrows, yellow-bellied sapsuckers, starlings, and mourning doves. They found a consistent pattern of lung congestion, edema and occasional hemorrhage among the dead birds, consistent with death from inhalation of PTFE fumes. The Department of Public Health attributed the bird deaths to fumes the factory, and negotiated a plan for emission reductions [16].
Killed: 80 to 100 wild birds
Implicated in death: Fumes from non-stick manufacturing facility
In 1997 scientists in England reported that 80 to 100 wild birds were found dead in a four-hour time span approximately 700 feet from an industrial plant in western England that coated sheet metal with non-stick paint containing PTFE. On the afternoon of the bird deaths, the plant had some problems that resulted in oven temperatures raised up to 880 degF (470 degC) to improve the coating of the sheet metal with the PTFE-containing paint. Wind direction was consistent with fumes travelling from the plant to where the dead birds were found. The authors write, "Most of the birds were found dead, but some were alive, gasping for breath, and died shortly afterwards" [15].
References
[1] Kreger Theresa 2003. "Teflon deaths." Email correspondence to EWG. April 2003.
[2] Birdhotline 2003. "Warnings to keep your bird safe." Accessed via the Internet at http:/www.birdhotline.com/comment.htm. May 2003.
[3] Shively Carol. 2003. "PTFE fumes kill family's pet birds!" Accessed online at www.quakerville.com/qic/ezine/96Issue5/qteflon.htm. April 2003.
[4] Talking Green Parrot Aviary. 2003. Information accessed online at www.tgpa.com. April 7 2003.
[5] Sehnal, D. 2003. Personal correspondence via email with Environmental Working Group. April and May 2003.
[6] Grahme 2003. "Teflon-related bird information." Email correspondence to Environmental Working Group. April 24 2003.
[7] Stewart Bob. 2002. Personal communication with Dr. Jennifer Klein, Environmental Working Group. May 9, 2002.
[8] Stewart Bob. 2002. Personal email communication with Anne Morgan, Environmental Working Group. [date]
[9] Anonymous 2003. Email correspondence to Environmental Working Group. April 2003.
[10] Anonymous 2003. Email correspondence to Environmental Working Group. April 2003.
[11] Hopkins, Steve 2001. “Bird deaths linked to Teflon coating.” Waikato Times. Hamilton, New Zealand. Independent Publishers Ltd. July 11, 2001. Copyright 2001 Independent Publishers Ltd.
[12] Blandford, TB., Seamon, PJ., Hughes, R., Pattison, M and Wilderspin, MP. 1975. A case of polytetrafluoroethylene poisoning in cockatiels accompanied by polymer fume fever in the owner. Vet Rec 96(8): 175-8.
[13] Boucher M, Ehmler TJ, Bermudez AJ. 2000. Polytetrafluoroethylene gas intoxication in broiler chickens. Avian Dis 2000 Apr-Jun;44(2):449-53.
[14] Zanen AL, Rietveld AP. 1993. Inhalation trauma due to overheating in a microwave oven. Thorax 48:300-2.
[15] Pennycott TW, Middleton JD. 1997. Suspected PTFE toxicity in wild birds. Vet Rec 141:255.
[16] Canadian Cooperative Wildlife Health Centre (CCWHC). 1997. “Suspected Teflon toxicosis in songbirds.” CCWHC Newsletter. Volume 4, Number 3, Summer 1997. Accessed online May 2003 at http://www.fas.org/ahead/news/ccwhc/summer97.htm#ont
[17] Daniels, Mary. 1986. “Stove fumes killing cages birds; overheating coated pans can bring quick death,” Chicago Tribune. March 9, 1986.
[18] Daniels, Mary. 1987. “Health debate; non-stick drip pans catch heat.” Chicago Tribune. March 29 1987.
[19] Vaughn, Sam. 1994. “What is a safe toy?” NCS Magazine. The National Cockatiel Society (NCS). Accessed online May 2002 at http://www.cockatiels.org/articles/behaviour/safetoys.html.
100 websites that discuss birds and Teflon toxicosis
upatsix.com
Description: Up At Six is a very comprehensive pet bird care website. Lists common household hazards and provides a search engine for research by topic or keywords. Also links to avian associations, aviaries, sanctuaries, shopping, online chat groups, and many other bird-related issues.
Association of Avian Veterinarians: Basic Bird Care
http://www.aav.org/basic_care.htm#Avoid
Description: Association of Avian Veterinarians promotes avian medicine and stewardship. Become a member, find an avian vet, learn about AAV programs and policy statements.
PetPlace.com article: Dangers in the Kitchen
http://www.petplace.com/articles/artShow.asp?artID=3534
Description: PetPlace.com offers access to a wealth of medical and wellness pet information. Features an archive of more than 1350 pet-related articles searchable through a user-friendly database.
The National Cockatiel Society: General Bird Care
http://www.cockatiels.org/articles/care/gencare.html
Description: The official website of the National Cockatiel Society. Features a question and answer bulletin board with scheduled chats and “reading rooms” of cockatiel-related articles.
Planet-Pets.com: Housing/Feeding Birds
http://www.planet-pets.com/feedbrd.htm
Description: Planet Pets serves the public by finding, evaluating, selecting, organizing, and offering quality pet-related information and resources. Features a free online newsletter, kids korner, ask an expert, links to services.
Birds and More
www.birdsandmore.com
Description: Online store for birds and their owners. Includes health tips and general care for your bird.
TalkToTheVet - Caged Bird Housing Considerations
http://www.talktothevet.com/ARTICLES/BIRDS/Pbirdhousing.HTM
Description: Talk To The Vet is an online veterinarian consultation service. Links to vets, free newsletter, pet-specific articles, etc.
Bird Crazy - Safety Guide for Plants and Poisons
http://www.birdcrazy.com/newsite/articles/plants.htm
Description: Bird Crazy is an Online aviary based in San Diego, CA. Comprehensive list of poisonous plants and household poisons and well as a list of safe plants.
Birds N' Ways eZine
http://www.birdsnways.com/wisdom/ww1e.htm
Description: Guide to parrots and other exotic pet birds. Offers dozens of links to shopping, chatting, information and miscellaneous bird-related sites. Offers access to thousands of articles searchable by keyword, topic, author and website.
ASPCA - Managing Pet Bird Toxicoses
http://www.aspca.org/site/Search?query=PTFE&inc=10
Description: American Society for the Prevention of Cruelty to Animals exists to promote humane principles, prevent cruelty and alleviate fear, pain and suffering in animals. Use the search engine (search “PTFE”) to find a downloadable report “Managing Pet Bird Toxicoses”.
Additional Links:
Starling Talk
http://www.starlingtalk.com/warning.htm
Parrot Parrot
http://www.parrotparrot.com/birdhealth/kola.htm
Exotic Pet Vet
http://www.exoticpetvet.net/about.html
Kaytee
http://www.kaytee.com/companion_animals/birds/health/
PETA
http://www.petauk.org/mc/facts/fsc16.html
Pet Bird Report
http://www.petbirdreport.com/articles/dangers.html
The Parrot Chronicles
http://www.parrotchronicles.com/departments/hazards.htm
Feather Care Aviary
http://home.cogeco.ca/~jjackson283/index.htm
The Parrot Society of Australia
http://www.parrotsociety.org.au/
Stanley’s Quakerville
http://www.quakerville.com/features/ptfe.asp
Lafeber Company
http://www.lafeber.com/docs_book/13/thirty_three_dangers.htm
The Aviary
http://theaviary.com/teflon.shtml
Avitec Exotic Birds
http://www.avitec.com/Birdcare.html
Budgie.org
http://www.budgies.org/info/teflon.html
Those Majestic Macaws!
http://www.exoticbird.com/teflon.html
Taking Care Of Your New Bird
http://www.ddc.com/~kjohnson/safety.htm
Lovebird Center
http://www.lovebirdcenter.com/7hazards.htm
Cockatiel Cottage
http://www.cockatielcottage.net/hazards.html
New England Exotic Bird Sanctuary
http://www.neebs.org/poisons.htm
South Bay Bird Society
http://www.sobaybirdsoc.com/hazards.htm
Austin Bird Club
http://www.austinbirdclub.com/dangerous.htm
Old World Aviaries
http://www.oldworldaviaries.com/text/styles/teflon.html
http://www.oldworldaviaries.com/text/miscellaneous/buying_a_bird.htm
Bignest Aviaries
http://www.bignest.com/food.htm
http://www.bignest.com/misgrey.htm
Wee B Toys
http://www.weebbirds.com/firstaid.htm
Pet Bird Care Safety Tips and Information
http://members.tripod.com/~DAdams/qkbrdinf.html
Parrot Online
http://www.parrotline.org/teflon.html
Bella Online
http://bellaonline.com/articles/art3973.asp
Senegal
http://www.makasnest.com/senegal/Teflon/toxins.htm
Feathers and Tails
http://www.feathersandtails.org/holiday.html
Birds By Donna
http://www.birdsbydonna.com/BIRDS_BY_DONNA/Basic_Bird_Cax.html
Kindness Club
http://www.kindnessclub.nb.ca/pets/BirdSafety.html
Rockport Roost
http://www.rockportroost.com/Birdsafetytips.htm
Animal Lovers Pet Shop
http://www.animalloverspetshop.com/whatsnew.htm
The Real Macaw
http://www.realmacaw.com/pages/tielcare.html
Wool-N-Wings
http://www.parrots.com/faq-hn.htm
African Lovebird Society
http://www.africanlovebirdsociety.com/lovebirdcare/
North American Cockatiel Society
http://www.cockatiel.org/articles/hazards.html
Angel Wings Aviary
http://www.parrot-breeder.com/parrothazards.htm
A&M Aviaries
http://www.petbirdbreeder.com/hazards.htm
The Animal Clinic of Clifton
http://www.pethealthcare.net/html/body_household_dangers.html
Birdsafe.com
http://www.birdsafe.com/story_bird33.htm
NetPets
http://www.netpets.org/birds/healthspa/poisons2.html
Arizona Avian Breeders Association
http://azavianbreeders.org/BirdCare.shtml
Avian Avenue
http://zachary.avianavenue.com/BirdSafety.html
Ask Dr. Petra
http://www.drpetra.com/Birds/Household%20Dangers.htm
Long Island Parrot Society of New York
http://www.liparrotsociety.org/harmful.htm
http://www.liparrotsociety.org/teflon!.htm
The Grey Place
http://www.africangreys.com/hints/emergency.htm
Birds Birds Birds!
http://parrotfeathers.tripod.com/care.htm
The Family Pet Show
http://www.familypetshow.com/toxocosis.html
Doctors Foster and Smith
http://www.drsfostersmith.com/Articles/bird_commoninjuries.cfm
Parrots Canada
http://www.parrotscanada.com/birdowner/forthenewbirdowner.html
Birds Just Wanna Have Fun
http://www.birdsjustwannahavefun.com/durability_info.htm
American Animal Hospital Association
http://www.healthypet.com/Library/prevent-18.html
Soldan’s Feed and Pet Supplies
http://www.soldanspet.com/petperspectives/bird_basics.htm
British Columbia SPCA
http://www.spca.bc.ca/Factsheets/birdcare.htm
The Avian Medicine Chest
http://www.petmedicinechest.com/avian/discussions/chemicalpoisoningtext.asp
Pet Parrot Club UK
http://www.petparrotclubuk.co.uk/help/hazards.htm
The Pet Bird Website
http://www.petbird.co.uk/housedangers.htm
Quaker Parakeet
http://www.quakerparakeet.com/b4ubuy.html
Houston Pet Talk
http://www.houstonpettalk.com/Column_Bird_Buddies.htm
Hex Aviary
http://www.hexaviary.com/hexlocal.htm
Birds of a Feather
http://www.boaf.com/article_11.htm
All Creatures Animal Hospital
http://www.all-creatures.com/care/bird.htm
Feathered Friends Forever
http://www.featheredfriendsforever.org/homesafety.html
Bird Crazy
http://www.birdcrazy.com/toxins.htm
The Parrotlet Parlour
http://www.theparrotletparlour.com/Parrotlet_Hazards.shtml
The Windhover Veterinary Center
http://www.windhovervet.com/birdcare.htm
Arizona Bird Supply
http://www.azbirdsupply.com/bird%20care_basics.htm
The PetCARE Information Center
http://petcare.umn.edu/OPets/Articles/BirdTox.htm
The Holistic Bird Newsletter
http://www.holisticbirds.com/HBN01/octnov/pages/chronic1.htm
Win Rose Animal Hospital
http://www.winrosevet.com/Birds.htm
Harmony’s World of Pets
http://www.petvets.com/petcare/birdscolumn.html
Janice’s Bird Haven
http://www.wtv-zone.com/JaniceMarr/feathered/Care.html
The Bird Hotline
http://www.birdhotline.com/vet.htm
The True Parrot
http://thetrueparrot.homestead.com/EnvironmentalNeeds.html
The Grey-cheeked Parakeet
http://www.lib.montana.edu/~bmarsh/cheeky/dosdonts.html
Dandy the African Grey Parrot
http://www.vdnent.com/html/articles/part_behav_be-a-bird.htm
Quaker Parrots
http://www.quakerparrots.com/qtips/more_thoughts_on_teflon.htm/
http://www.quakerparrots.com/qtips/dangers_of_teflon.htm
Animal Rescue League of Iowa
http://www.arl-iowa.org/PetCare/Birds.asp
Wasatch Avian Education Society
http://www.wasatchavian.com/fosterguide.html
Dane County Humane Society
http://www.humanesociety.dane.wi.us/care/bird_groom.html
Montreal Bird and Exotic Animal Hospital
http://www.birdandexoticvet.com/avian.html
Parrot Passions UK
http://www.parrotpassionsuk.com/Advice/hazards.htm
Birdlife
http://www.birdlife.com/care/dangers.html
Pretty Birds
http://www.prettybirds.net/Parrotsafehome.htm
The Animal Network
http://www.animalnetwork.com/birds/library/articleview3.asp? Section=&RecordNo=2721
Pet Finder
http://www.petfinder.org/journalindex.cgi? path=/public/animalcare/bird/1.3.404.txt
Budgies ’N Tiels
http://www.budgies-n-tiels.com/contents/health.html
Pair o' Keets and Qweak n' Tails
http://www.geocities.com/monicaarnouk/BirdHazards.htm
SunConure
http://www.sunconure.com/safety.html
Give Us A Home
http://www.giveusahome.co.uk/birds/faq.htm
Eve’s India
http://www.evesindia.com/family/pets/bird_safety.html
MSN Health
http://content.health.msn.com/content/article/35/1728_84136
Pet Education
http://www.peteducation.com:80/article.cfm?cls=15&cat=1829&articleid=2418 http://www.peteducation.com:80/article.cfm?cls=15&cat=1912&articleid=1559
North Fork Animal Hospital
http://www.northforkanimalhospital.com/animals/birds/birds_quaker.htm
The government fails to study Teflon's safety

The government has never rigorously studied the safety of Teflon pans. The Food and Drug Administration approved Teflon as a Food Contact Substance in 1960 after reviewing a hamburger frying study in which elevated levels of fluorinated compounds were found in the hamburger fried on Teflon. FDA judged these levels to be of little health significance. After a prominent Midwestern paper reported on cases of bird deaths and human polymer fume fever, the Consumer Product Safety Commission stated that the Commission would investigate only upon receiving a formal complaint with underlying data. CPSC failed to ban Teflon-coated drip pans, which can reach 1000°F, even after well-publicized poisoning incidences.

References
[1] Blumenthal, Dale. 1990. "Is that newfangled cookware safe?" FDA Consumer. October 1990.
Tips for bird owners & the rest of us
Bird owners should avoid non-stick cookware. It is not enough to keep the bird in a room of the house far from the kitchen. In numerous cases, Teflon kitchen fumes have killed birds far from the source, in other rooms of the house. Bird owners should also avoid other heated appliances coated with non-stick surfaces, including waffle irons, griddles, stovetops and ovens, space heaters, irons and ironing board covers. Many of these are also implicated in deaths of pet birds by Teflon toxicosis.
What about the rest of us?
Even if you don't have pet birds, Teflon toxicosis is something to be aware of and, in our view, concerned about. There is absolutely no doubt that when heated to temperatures well within normal cooking range, for instance, Teflon and products with other non-stick PFC coatings emit toxic fumes that can be harmful to people. They can make you and your family sick. The long term effects of routine exposure to Teflon fumes, and "fume fever" itself, have not been adequately studied.
Our advice: phase out the use of Teflon or non-stick cookware in your home. In fact, avoid any kitchen equipment that contains Teflon or other non-stick components that are heated to high temperature during use. It's not always easy to find out about Teflon components in ovens, space heaters or other devices, and in fact, some bird owners were reassured their products were Teflon-free, only to find out the truth when their birds were killed. That's why we want the government to require labels on such products. In the meantime, you should inquire before purchase or continued use.
In addition to avoiding risks to you and your family, phasing out Teflon and other non-stick products that might be heated to high temperatures will help reduce the impacts of the "PFC" economy on human health and wild animals. (See our Web site, PFCs: Global Contaminants).
Tips on safe cookware
When heated, cookware coated with Teflon and other non-stick surfaces emits fumes that can kill birds and potentially sicken people. You can avoid exposures to the fumes from Teflon and other non-stick cookware by phasing out your home use of these products. If you can afford to replace your non-stick cookware now, do so.
Statistics reported by the Cookware Manufacturers Association indicate that 90 percent of all the aluminum cookware sold in the United States in 2001 was coated with non-stick chemicals like Teflon (Cooks Illustrated, September 2002). Chemicals and tiny, toxic Teflon particles released from heated Teflon kill household pet birds. At least four of these chemicals never break down in the environment, and some are widely found in human blood. Consumers concerned about the effects of Teflon on human health and the environment should consider these alternatives:
Stainless Steel
Stainless steel is a terrific alternative to a non-stick cooking surface. Most chefs agree that stainless steel browns foods better than non-stick surfaces. In their 2001 review of sauté pans, Cooks Illustrated, an independent publication, chose a stainless steel pan over otherwise identical non-stick models. They also recommended stainless steel pan roasters over non-stick.
Cast Iron
Cast iron remains a great alternative to non-stick cooking surfaces. Lodge, America’s oldest family-owned cookware manufacturer, refers to their cookware as “natural non-stick.” Cast iron can be pre-heated to temperatures that will brown meat and will withstand oven temperatures well above what is considered safe for non-stick pans. Cast iron is extremely durable and can now be purchased pre-seasoned, ready-to-use.
Other Cooking Surfaces
Because Teflon coated non-stick surfaces fail to brown foods there has been a push to find other “non-stick” cookware coating that will allow the use of higher temperatures and still clean up easily. Some examples include ceramic titanium and porcelain enameled cast iron. Both of these surfaces are very durable, better at browning foods than PTFE (Teflon) non-stick coatings, and are dishwasher safe. In her New York Times piece, "In Search of a Pan That Lets Cooks Forget About Teflon," Marian Burros recommends Le Creuset enameled cast iron pans with a matte black interior. Anodized aluminum is another alternative, but some people question its safety, citing evidence in some studies linking aluminum exposures to Alzheimers.






